
Materials Science, Vol.
46, No.
1, 2010
INFLUENCE OF POLYACRYLIC ACID ON THE PROTECTIVE PROPERTIES
OF THE KORSOL INHIBITOR
Z. V. Slobodyan,
1
L. A. Mahlatyuk,
1,2
and R. B. Kupovych
1
UDC 620.197.3
Polyacrylic acid (PAA) is a dispersant of hardness salts. Its anticorrosion properties are better than the
anticorrosion properties of the dispersants produced by the Nalko Firm. In the KORSOL composition, it
does not reveal the synergetic action but does not deteriorate the protective properties of the inhibitor.
Keywords: corrosion rate, depth index, degree of protection, corrosion inhibitor, dispersant, polyacrylic
acid.
The application of water-soluble polymers in inhibiting compositions for neutral media is realized for two
purposes. On the one hand, they act as corrosion inhibitors and, on the other hand, as dispersants of hardness
salts deposited on the metal surfaces [1]. It is known that the solutions of high-molecular compounds are ly-
ophilic thermodynamically stable and reversible systems [2]. This is why the inhibitors containing polymeric
electrolytes also have stable physicochemical characteristics.
The KORSOL inhibitor is a well-known reagent, which efficiently inhibits corrosion and scaling [3]. Note
that, in the case of its long-term application, old salt deposits are washed out [4]. However, this process is quite
slow and insufficiently efficient. At the same time, by dispersing the deposits, polymeric electrolytes improve the
hydrodynamic and heat-exchange characteristics of heating networks, which is especially important for systems
whose depreciation period is already exhausted. However, in this case, it is necessary to guarantee that these sub-
stances do not weaken the anticorrosion action of the inhibitors.
Polyacrylic acid (PAA)
[ CH
2
|
CH]
n
COOH
is known as a substance with moderate dispersing action [5]. However, its anticorrosion properties in waters
with various levels of hardness are studied quite poorly. Its influence on the efficiency of the KORSOL inhibi-
tor has not been investigated yet [3]. This is why we analyze the influence of PAA on the protective properties
of the inhibitor in waters with various contents of hardness salts.
Materials and Procedures
We tested specimens of 20 steel (with a diameter of 22 mm and
h
= 1.5 mm). The KORSOL inhibitor
(d = 1.52 g/cm
3
)
was studied at concentrations of 40–
200 mg/dm
3
, PAA (with a molecular mass of 100,000)
1
Karpenko Physicomechanical Institute, Ukrainian National Academy of Sciences, Lviv, Ukraine.
2
Corresponding author; e-mail: [email protected].
Translated from Fizyko-Khimichna Mekhanika Materialiv, Vol.
46, No.
1, pp.
123–125, January–February, 2010. Original article submit-
ted April 20, 2009.
1068-820X/10/4601–0139 © 2010 Springer Science+Business Media, Inc. 139

140 Z. V. SLOBODYAN, L. A. MAHLATYUK, AND R. B. KUPOVYCH
was studied at concentrations of 20–
80 mg/dm
3
, and the Na7302
(d = 1.46 g/cm
3
)
and Na8510
(d =
1.49 g/cm
3
)
dispersants produced by the Nalko Firm were analyzed at concentrations of 5–100
mg/dm
3
. Dif-
ferent contents of calcium carbonate were obtained by varying the ratio of
CaCl
2
to
NaHCO
3
. The concentra-
tion of
Ca
2+
was determined by trilonometric titration with murexide [6]. The corrosion rate was evaluated by
the weight method. The degree of protection is given by the formula
Z =
K
m
K
m
K
m
100%
,
where
K
m
is the rate of corrosion in the medium and
K
m
is the corrosion rate in the presence of the inhibitor.
Results and Discussion
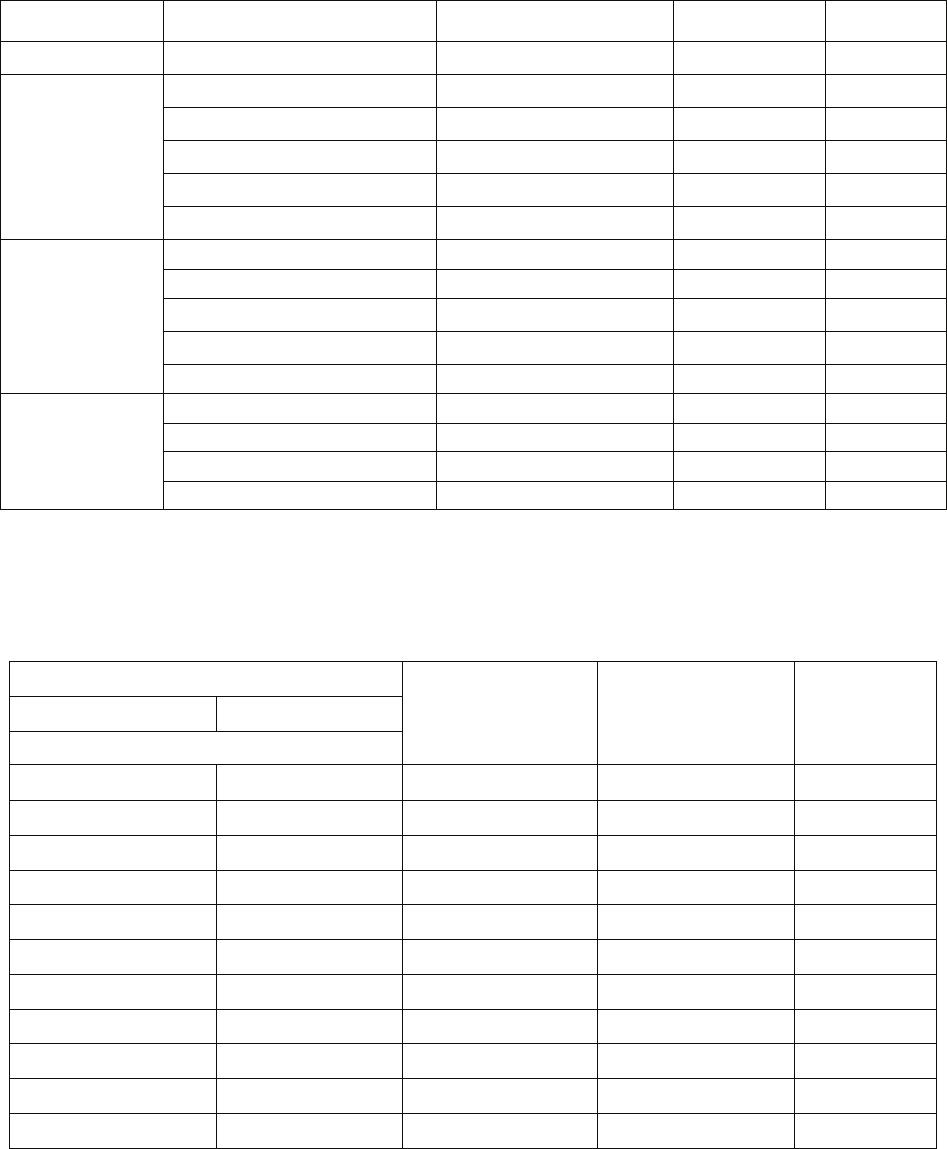
Within the range of the investigated concentrations, as the content of calcium ions in water increases, the
rate of corrosion in steel decreases (Table 1). This regularity is connected with the moderate formation of a pro-
tective film of calcium carbonate on the surfaces of the specimens. The results of testing of the inhibitor in wa-
ters with different levels of hardness (Table 1) did not revealed any direct relationship between its efficiency and
the concentration of calcium ions. The maximum degrees of protection were recorded at concentrations of
0.01
g- ion/dm
3
and 0.001
g- ion/dm
3
. This is why the inhibiting properties of dispersants were investigated
in water containing 0.0135
g- ion/dm
3
Ca
2+
. Within the concentration range 5–100
mg/dm
3
, the Na7302 and
Na8510 dispersants slightly increase the corrosion resistance of 20 steel but, within the range
50–80
mg/dm
3
,
they even somewhat accelerate the corrosion processes (Table 2).
Table 1. Influence of the Concentration of
Ca
2+
in Water in the Presence of the KORSOL
Inhibitor
(200 mg/dm
3
)
on the Corrosion Rate
(K
m
)
, Depth Index
(
)
,
and the Degree of Protection
(Z)
of 20 Steel
Concentration,
g/dm
3
CaCl
NaHCO
3
Concentration
[Ca
2+
]
,
g- ion/dm
3
K
m
10
2
,
g/(m
2
h)
,
mm/yr
K
m
10
3
,
g/(m
2
h)
, mm/yr
Z
,
%
1.5 1.5 0.0135 5.17 0.058 5.8 0.06 88
1.2 1.2 0.0108 8.19 0.091 4.4 0.005 95
1.0 1.0 0.0090 9.48 0.106 8.8 0.0096 91
0.7 0.7 0.0063 10.34 0.115 8.8 0.009 91
0.5 0.5 0.0045 10.99 0.122 11.0 0.12 90
0.2 0.2 0.0018 11.21 0.125 4.4 0.004 96
0.05 0.05 0.00045 12.13 0.135 16.0 0.017 84
INFLUENCE OF POLYACRYLIC ACID ON THE PROTECTIVE PROPERTIES OF THE KORSOL INHIBITOR 141
Table 2. Influence of the Na7302 and Na8510 Dispersants and PAA on the Corrosion Rate
(K
m
)
,
Depth Index
(
)
, and Degree of Protection
(Z)
of 20 Steel in Water
(the Concentration
[Ca
2+
]
Is Equal to
1.3
10
2
g- ion/dm
3
)
Medium
Concentration,
mg/dm
3
K
m
10
2
,
g/(m
2
h)
, mm/yr
Z
, %
H
2
O
– 10.98 0.1098 –
5 10.66 0.119 2.9
25 9.71 0.108 11.6
50 10.98 0.122 0
80 11.14 0.124 –1.46
Na7302
100 10.82 0.120 1.45
5 10.51 0.117 4.3
25 10.03 0.112 8.7
50 11.3 0.126 –2.9
80 10.35 0.115 5.7
Na8510
100 10.35 0.115 5.7
5 9.2 0.102 16.2
40 8.6 0.095 20.9
80 8.7 0.097 20.7
PAA
100 8.8 0.098 19.8
Table 3. Corrosion Rate
(K
m
)
, Depth Index
(
)
, and Degree of Protection
(Z)
of 20 Steel in Water for
Different Ratios of PAA and KORSOL (the Concentration
[Ca
2+
]
Is Equal to
1.3
10
2
g- ion/dm
3
)
Medium
KORSOL PAA
mg/liter
K
m
10
2
,
g/(m
2
h)
, mm/yr
Z
, %
Water – 7.86 0.088 –
40 – 1.98 0.023 75
80 – 1.97 0.022 75
160 – 0.94 0.0010 88
200 – 0.59 0.0065 92
80 20 1.76 0.0196 78
120 40 1.27 0.014 84
40 40 2.40 0.082 69
80 80 1.96 0.022 75
160 40 1.06 0.012 87
200 40 0.82 0.009 90
142 Z. V. SLOBODYAN, L. A. MAHLATYUK, AND R. B. KUPOVYCH
The anticorrosion action of PAA is insignificant within the concentration range 5–100
mg/dm
3
but more
pronounced than the anticorrosion action of the Na7305 and Na8510 dispersants. The degree of protection of 20
steel is practically independent of the concentration of acid. Therefore, the mixtures of PAA with the KORSOL
inhibitor were studied for the concentrations of PAA varying within the range 20–80
mg/dm
3
(Table 3). In this
case, the efficiency of the inhibitor insignificantly increases or even somewhat decreases for low concentrations
(40
mg/dm
3
).
Thus, PAA does not exhibit noticeable synergetic properties in compositions with the KORSOL inhibitor.
For the investigated concentrations, the efficiency of corrosion protection is determined by the concentration of
the inhibitor (Table. 3). As a dispersant, PAA can be used in mixtures with the KORSOL inhibitor without de-
creasing the efficiency of corrosion protection.
REFERENCES
1. A. I. Altsybeeva and S. Z. Levin, Inhibitors of Corrosion in Metals [in Russian], Khimiya, Leningrad (1968).
2. D. A. Fridrikhsberg, A Course of Colloid Chemistry [in Russian], Khimiya, Leningrad (1974).
3. Z. V. Slobodyan, D. M. Zaverbnyi, V. M. Zhovnirchuk, et al, Inhibitor of Corrosion and Scaling [in Ukrainian], Patent of Ukraine.
UA 20906A, MPK
7
C23F 11/08, Published on 27.02.98, Bull. No. 1.
4. Z. Slobodyan, H. Nykyforchyn, and L. Mahlatyuk, “Specific features of the protective action of inhibitors of the KORSOL family
under the conditions of corrosion, biocorrosion, and corrosion-mechanical fracture of steels,” Fiz.-Khim. Mekh. Mater., Special Is-
sue, No. 7, 533–537 (2008).
5. A. D. Zimon, Colloid Chemistry [in Russian], Agar, Moscow (2003).
6. A. K. Babko and I. V. Pyatnitskii, Quantitative Analysis [in Russian], Vysshaya Shkola, Moscow (1968).
