Mat. Res. Soc. Symp. Proc. Vol. 621 © 2000 Materials Research Society
R3.5.1
A Novel Approach for True Work Function Determination of Electron-Emissive Materials
by Combined Kelvin Probe and Photoelectric Effect Measurements
Bert Lägel, Iain D. Baikie, Konrad Dirscherl and Uwe Petermann,
Department of Applied Physics, The Robert Gordon University, Aberdeen, UK.
ABSTRACT
For the development of new electron-emissive materials knowledge of the work function (φ)
and changes in φ is of particular interest. Among the various methods, the ultra-high vacuum
(UHV) compatible scanning Kelvin Probe has been proven to be a superior technique to measure
work function changes due to e.g. UHV cleaning processes, chemical contamination, thermal
processing etc. with high accuracy (<1meV).
The Kelvin Probe measures local work function differences between a conducting sample and
a reference tip in a non-contact, truly non-invasive way over a wide temperature range. However,
it is an inherently relative technique and does not provide an absolute work function if the work
function of the tip (φ
tip
) is unknown.
Here, we present a novel approach to measure φ
tip
with the Kelvin Probe via the photoelectric
effect, where a Gd foil is used as the photoelectron source. This method thus provides the true
work function of the sample surface with an accuracy of approx. 50meV. We demonstrate the
application of the technique by in situ work function measurements on evaporated layers of the
low work function material LaB
6
on a Re substrate and follow the changes in φ of LaB
6
due to
the surface adsorption of residual gas molecules. Thus, the extended Kelvin Probe method
provides an excellent tool to characterise and monitor the stability of low work function surfaces.
INTRODUCTION
The work function is an extremely sensitive indicator of changes in surface and interface
chemical composition, adsorbate induced surface dipole and surface roughness. Further, for the
characterisation of electron emissive materials, the work function is one of the most important
parameters and low φ materials are of particular interest. However, due to the adsorption of
residual gases, the work function of these materials is known to increase with time, adversely
affecting their emission properties.
The Kelvin Probe [1, 2] can be used to monitor these work function changes in a non-contact,
non-invasive way with high accuracy (< 1meV) [3, 4] up to temperatures of approx. 900K. It has
been utilised e.g. for thin film studies [5, 6, 7], characterisation of oxides and thin films [8],
semiconductor surface processing [9] and surface charge imaging [10], investigation of the
adsorption kinetics of oxygen on Si(111) surfaces [11] as well as for biological applications [12].
However, the Kelvin Probe is an inherently relative technique as it measures the average
work function or contact potential difference (CPD) between a vibrating reference electrode (the
tip) and the surface under investigation. Thus, in order to obtain the absolute value of the work
function of the specimen, it is necessary to know the work function of the reference electrode,
φ
tip
. This could be determined e.g. by a CPD measurement on a clean reference surface. The
accuracy of this method however depends on tabulated φ values, which are often valid only for
the given experimental setup and the assumption of a clean reference surface.
R3.5.2
We have developed a relatively simple extension of the technique as an approach for a more
reliable method to determine φ
tip
. This is achieved via measurement of the current voltage (I-V)
characteristic of the photoelectric emission from a low work function surface, such as Gd,
illuminated by monochromatic light of a fixed wavelength as discussed e.g. in [13]. The work
function of any conductor or semiconductor can then be obtained by subsequent Kelvin Probe
CPD measurements.
We have utilised this technique to investigate low work function materials as possible targets
for use in Hyperthermal Surface Ionisation (HSI), a new mass spectroscopy ionisation technique
which offers high ionisation efficiency and low amounts of cracking products [14, 15].
Lanthanum Hexaboride (LaB
6
) is particularly interesting in this respect because of its low work
function and its surface re-activation capabilities after adsorption of residual gases via flash-
annealing [16].
EXPERIMENTAL METHOD
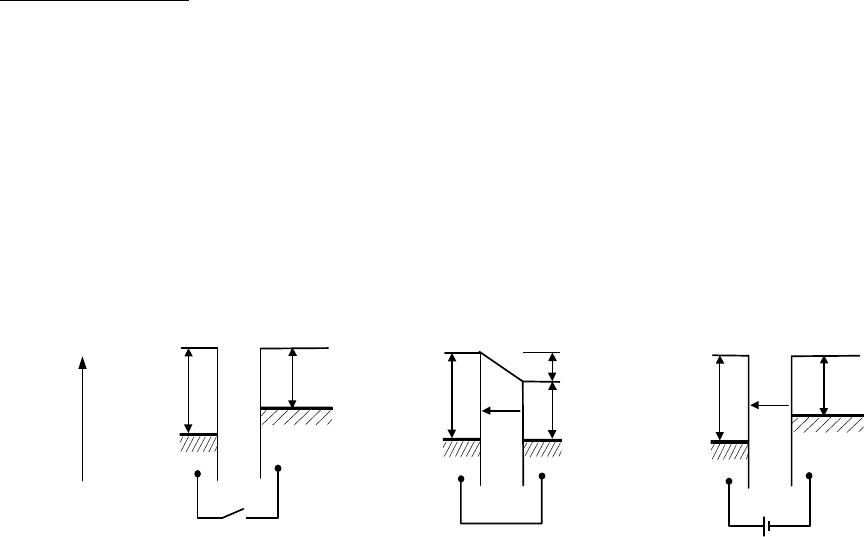
The Kelvin Method
The Kelvin Probe consists of a flat circular electrode (reference electrode, tip) suspended
above and parallel to a stationary electrode (the specimen, S), thus creating a simple capacitor. If
an external electrical contact is made between the two electrodes their Fermi levels equalise and
the resulting flow of electrons from the metal with the lower work function produces a contact
potential difference, V
CPD
, between the plates as shown in fig. 1(b):
eV
CPD
= φ
S
− φ
tip
, (1)
where e is the electronic charge and φ
tip
and φ
S
are the work functions of the tip and sample,
respectively. By vibrating the probe, a varying capacitance is produced which causes a current to
flow back and forth between the plates.
E
=
0
E
≠
0
− +
− +
− +
− +
Electronic Energy
(a)
E
Vac
φ
tip
ε
tip
φ
S
ε
S
V
b
= -V
CPD
(c)
φ
tip
ε
tip
φ
S
ε
S
eV
CPD
(b)
φ
tip
ε
tip
φ
S
ε
S
Figure 1. Electron energy level diagrams of two different metals (a) without contact, (b) with
external electrical contact, where E indicates the electrical field between the plates, and (c) with
inclusion of the backing potential.
ε
tip
and
ε
S
refer to the Fermi levels of the tip and sample,
respectively.
Inclusion of a variable “backing potential” V
b
in the external circuit permits biasing of one
electrode with respect to the other: at a unique point, where
CPDb
VV −= , the electrical field
between the plate vanishes, see fig. 1(c), resulting in a null output signal. The work function
difference between the electrodes is thus equal and opposite to the DC potential necessary to
produce a zero output signal.
R3.5.3
The CPD measurements were performed with a UHV compatible Kelvin Probe that
incorporates advanced “off-null” detection, where the balance point is determined by linear
extrapolation rather than nulling with a resolution of <1mV as well as automatic control of the
tip-to-sample spacing, which is an important requirement for accurate measurements [4, 17].
Measurements can be performed with a frequency of up to 1 data point per second.
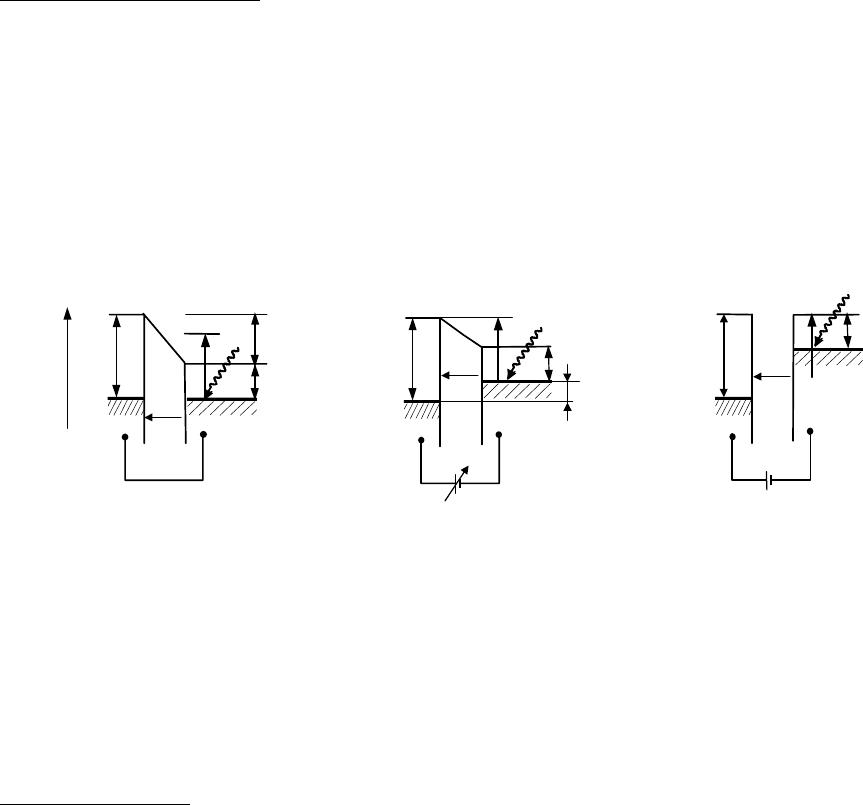
Photocurrent Measurement
In the photoelectric measurement mode the capacity of the arrangement is kept constant. The
photoelectrons emitted from a low work function surface (S) illuminated by monochromatic light
of energy E
ph
= hν with φ
tip
> hν > φ
S
are collected by the Kelvin Probe tip and measured as a
function of the applied backing potential V
b
(see figure 2). It can be seen that, at the onset of the
photocurrent collected at the Kelvin Probe tip, (fig. 2b), the work function of the tip equals the
photon energy E
ph
plus a required bias potential supplied by V
b
, i.e.,
btip
Vh +=
νφ
(onset). (2)
h
ν
E
≠
0
E
≠
0
E
=
0
Electronic Energy
(a)
φ
S
ε
S
eV
CPD
φ
tip
ε
tip
E
Vac
φ
S
ε
S
φ
tip
ε
tip
eV
b
h
ν
V
b
= V
b
(onset)
(b)
φ
tip
ε
tip
ε
S
V
b
= -V
CPD
φ
S
(c)
h
ν
Figure 2. Energy level diagrams of the Kelvin Probe in photocurrent measurement mode with
φ
tip
>
h
ν
>
φ
S
: (a) no photoelectrons are collected at the tip, (b), the onset of the photocurrent is
measured where
φ
tip
= h
ν
+ V
b
(onset) and in (c) the saturation current where
CPDb
VV −= .
With increasing bias potential, analogous to the Fowler theory of photoemisson from metals
near threshold [18], the photocurrent increases with the square of V
b
: I
ph
∝ V
b
2
. When V
b
equals
-V
CPD
(fig. 2c), electrons emitted from energy states below the Fermi level with virtually zero
velocity are only just able to reach the collector and the photocurrent will saturate.
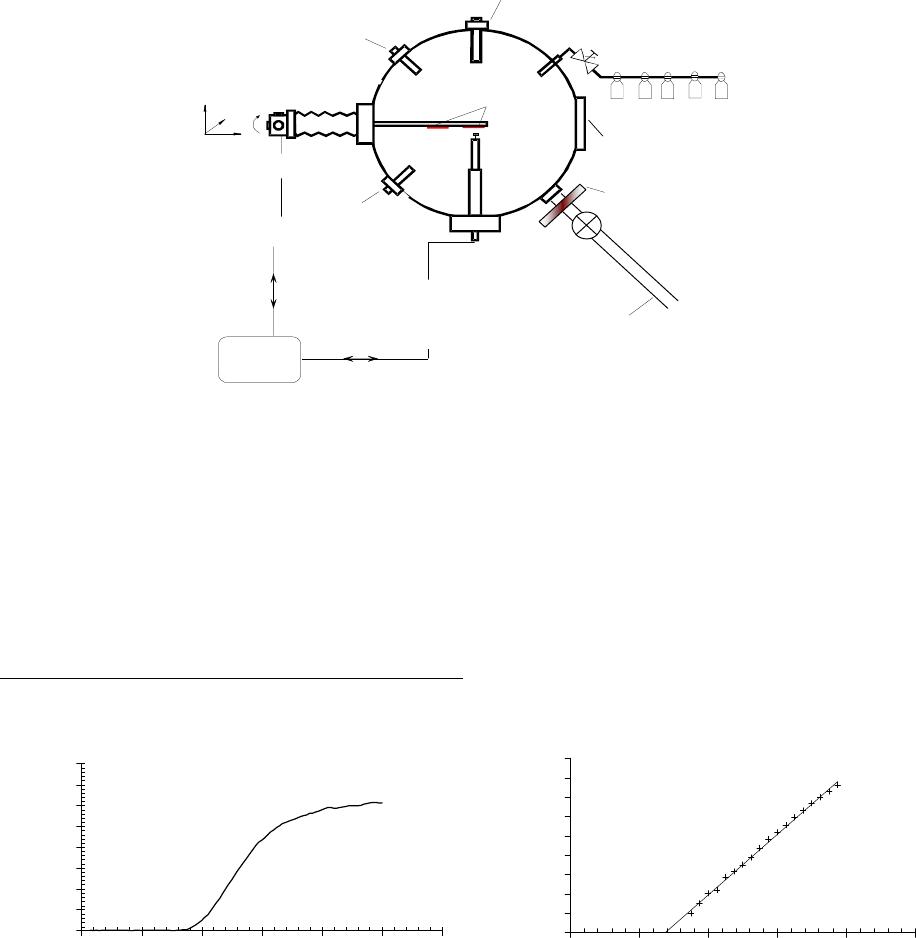
Experimental Setup
The experiments were carried out in a UHV chamber as shown in fig. 3 with a base pressure
of <4×10
-9
Torr. A polycrystalline gadolinium foil (25×12×0.1mm
3
) was used as the
photoelectron source. The sample was cleaned by repeated resistive heating up to 1200K for
several minutes. Gd has the advantage of having a relatively low work function of 3.1eV [19] and
is available in form of thin foils. Further, we found that the work function of Gd remained stable
within 100meV over several days. For photoemisson measurements the Gd foil was illuminated
by a 100W Hg-Cd-Zn spectral lamp through a sapphire viewport which is transparent for
wavelengths down to 200nm. The 312.6nm Hg emission was selected by a narrowband
interference filter (FWHM = ± 20nm).
R3.5.4
LaB
6
(purity 99%) films were generated via evaporation onto a polycrystalline rhenium foil
(25×12×0.025mm
3
, purity 99.99%). The foil was cleaned by repeated flash annealing to
temperatures above 2000K. The work functions of the samples were determined by first
obtaining φ
tip
of the Kelvin Probe via the photoeffect I-V measurement and a subsequent Kelvin
Probe CPD measurement of the respective sample. The UHV system was darkened during the
measurements and all filaments were extinguished.
Host
Computer
KP / SKP PE
Probe Oscillator
Pre-Amplifier
AD Converter
DA converter
I-V Converter
Pre-Amplifier
X-Y-Z Translation Stage
Fast Entry
Load Lock
X
Y
Z
Gas Inlet
System
Kelvin Probe
Samples
Stepper Motor
Control Unit
Ar O
2
H
2
CO
2
N
2
Quadrupole Mass
Analyser
Ion Gauge
Pulsed Nozzle Valve
Optical Rail
Bandpass Filter
(312.6nm)
Hg, Zn, Cd
Spectral Lamp
e-Beam Evaporator
Figure 3. Schematic of the UHV system with its associated peripherals and facilities
For CPD/photocurrent measurements the signal collected from the stainless steel tip
(Ø2.5mm) is converted into a voltage signal by a high gain (10
7
), low noise operational amplifier,
filtered and digitised by the AD-converter for further signal processing by the host PC. The
computer program controls tip oscillation and backing potential. This design thus provides a fully
automated measurement system.
RESULTS AND DISCUSSION
Kelvin Probe Tip Work Function Determination
Figure 4 shows typical data of the photoelectron emission from the Gd surface.
Figure 4. Typical photoemission data used to determine the work function of the Kelvin Probe:
(a) the current-voltage (I-V) curve and (b) the plot I
ph
1/2
versus bias voltage. Note that only data
from the linear portion of the curves are included in the fit.
0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
0 0.4 0.8 1.2 1.6 2
V
b
/ V
I
ph
1/2
/ a.u.
0
0.2
0.4
0.6
0.8
1
1.2
1.4
1.6
-1012345
V
b
/ V
I
ph
/ nA
(a) (b)
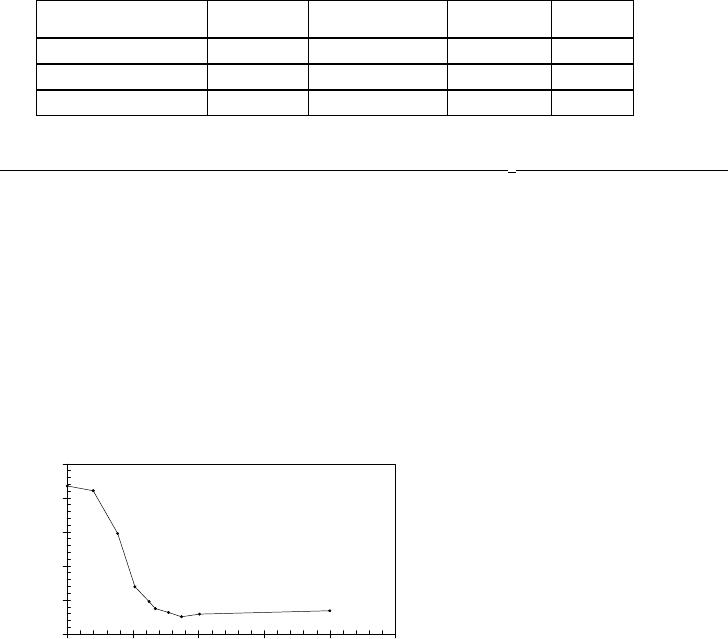
R3.5.5
In order to determine the onset of the photocurrent I
ph
, the square root of I
ph
is plotted versus
the applied bias voltage V
b
(fig 4b) and a least square fit of the linear part of the curve is
extrapolated to zero current. φ
tip
and φ
Sample
are then calculated via equations 1 and 2,
respectively. The results of the measurements are summarised in table I. The uncertainty of the
extrapolation is approx. ± 20mV. Further, taking the measurement error due to the thermal
energy distribution into account (26mV at 300K) and small changes in tip work function during
the measurement (<20meV), we estimate the overall error of the work function measurement to
be ± 50meV.
The work function determined for Gd (3.075 ± 0.050)eV
is, within the measurement error, in
agreement with that of (3.1 ± 0.150)eV
given in [19]. The work function measured for the clean
Re substrate was used as a calibration point for the absolute work function scale in fig. 5.
Table I. Results of the measurements to determine tip and sample work functions.
Measurement E
ph
/ eV V
b
(onset) / V V
CPD
/ V
φ
φφ
φ / eV
Kelvin Probe tip 3.966 0.551± 0.016
/
4.518
Gd
/
/
-1.443 3.075
Re
/
/
0.653 5.171
Application to Work Function Measurements of LaB
6
on Polycrystalline Re
In order to characterise the LaB
6
evaporation process, the work function change of the
Re/LaB
6
system was measured at different stages of the evaporation as shown in figure 5. After
an initial rapid decrease the work function changes only in small steps with further evaporation,
gradually achieving approx. 3.3eV. However, if the surface is ‘activated’ by flashing it to
1500°C, a further work function decrease to 2.536eV is obtained. The necessity for such an
activation procedure might be caused either by contamination or, indeed, a rearrangement of the
LaB
X
structure since its work function is known to depend strongly on the surface stoichiometry
of the compound [20].
The work function increase due to the adsorption of residual gas molecules of the LaB
6
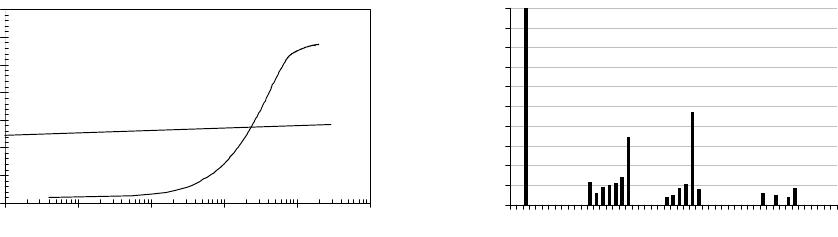
surface after activation and, for comparison, Gd, is shown in figure 6. With respect to the LaB
6
surface, we clearly observe 2 adsorption stages. The first one, marked AB, with an almost
constant gradient above 10L and the second one, after approx. 80L, with a lower and rather
continuous gradient leading to a work function of 3.65eV after an exposure of 200L.
Figure 5. Work function change
during evaporation of LaB
6
onto a
Re foil held at 300K. The current
onto the sample due to the partly
ionised molecular beam was used as
a measure of the particle flux.
3
3.5
4
4.5
5
5.5
0 5 10 15 20 25
particle flux / a.u.
φ(
φ(φ(
φ(
Re/LaB
6
)
/
eV
R3.5.6
This shows that LaB
6
is rather easily poisoned at 300K by the adsorption of the residual gas,
the largest component of which is molecular hydrogen. In contrast, although having a higher
initial work function, φ(Gd) increases by only 70meV after 200L exposure.
Figure 6. (a) Work function increase of LaB
6
and Gd due to the adsorption of residual gas
molecules at 300K. (b) The relative composition of the residual gas envelope at a base pressure
of 3.7
×
10
-9
Torr.
CONCLUSIONS
We have demonstrated the feasibility of a novel approach to determine the work function of
the reference tip of a Kelvin Probe via photoelectric current measurements on Gd using a
minimum of additional instrumentation. We found that the work function of Gd remains stable
over a prolonged period of time in the upper UHV pressure region of approx. 4×10
-9
Torr and is
therefore best suited for this purpose. The measurement error is estimated to be ± 50meV.
Using the new technique we have followed the evaporation of LaB
6
onto a polycrystalline Re
substrate. After activation, the surface work function at 300K was (2.536 ± 0.050) eV, increasing
to (3.65 ± 0.050) eV after an exposure of 200L to the residual gas. This indicates a very high
sensitivity of the work function of LaB
6
to the adsorption of gas molecules which may restrict its
use for applications requiring a stable low work functions surface at low temperatures.
ACKNOWLEDGEMENTS
This work was supported by EPSRC and DERA.
REFERENCES
1. Lord Kelvin, Philos. Mag
46
, 82 (1898)
2. W.A. Zisman.
Rev. Sci. Instrum.
3
, 367 (1932)
3. I.D. Baikie, K.O. Vanderwerf, H. Oerbekke, J. Broeze, A. Vansilfhout, Rev. Sci. Instrum.
60
,
930 (1989)
4.
I.D. Baikie, P.J. Estrup, Rev. Sci. Instrum.
69
, 3902 (1998)
5. M. Schmidt, H. Wolter, M. Nohlen, K.Wandelt, J. Vac. Sci Technol.
A12
, 1818 (1994)
6. E. Kopatzki, H.G. Keck, I.D Baikie, J.A. Meyer, ,R.J. Behm, Surf. Sci.
345
, L11 (1996)
7. I.D. Baikie, U. Petermann, B. Lägel, Surf. Sci.
433-435
, 770 (1999)
0
10
20
30
40
50
60
70
80
90
100
0 3 6 9 12 15 18 21 24 27 30 33 36 39 42 45 48
mass / a.m.u.
relative composition / %
2.5
2.7
2.9
3.1
3.3
3.5
3.7
3.9
0.01 0.1 1 10 100 1000
Exposure / Langmuir
φ
φφ
φ
(Re/LaB
6
) / eV
A
B
C
LaB
6
Gd
(a) (b)
R3.5.7
8.
I.D. Baikie, G.H Bruggink in Materials Reliability in Microelectronics III, edited by K.P. Rodbell,
W.F. Filter, H.J. Frost, P.S. Po,
(Mater. Res. Soc. Proc.
309
, Pittsburgh, PA, 1993), pp. 35-40
9.
I.D. Baikie in Chemical Perspectives of Microelectronic Materials II, edited by L.V. Interrante, K.F.
Jensen, L.H. Dubois, M.E. Gross
(Mater. Res. Soc. Proc.
204
, Pittsburgh, PA, 1991), pp. 363-368
10. B. Lägel , I.D. Baikie, U. Petermann in Defect and Impurity Engineered Semiconductors and
Devices II, edited by S. Ashok, J. Chevallier, K. Sumino, B.L. Sopori, W. Goetz, (Mater. Res.
Soc. Proc.
510
, Pittsburgh, PA, 1998), pp. 619-625
11. U. Petermann, I.D. Baikie, B. Lägel, Thin Solid Films
343-344
, 492 (1999)
12.
I.D. Baikie, P.J.S. Smith, D.M. Porterfield, P.J. Estrup, Rev. Sci. Instrum.
70
, 1842 (1999)
13. L. Apker, E. Taft, J. Dickey, Phys. Rev.
73
, 46 (1947)
14. A. Danon, A. Amirav, Isr. J. Chem.
29
, 443 (1989)
15. A. Danon, A. Amirav, Int. J. Mass Spectrom. Ion. Processes
96
, 139 (1990)
16. J.M. Lafferty, J. Appl. Phys.
22
, 299 (1951)
17.
I.D. Baikie, S. Mackenzie, P.J.Z. Estrup and J.A. Meyer, Rev. Sci. Instrum.
62
, 1326 (1991)
18. R.H. Fowler, Phys. Rev.
33
, 45 (1931)
19. D.E. Eastman, Phys Rev.
B2
, 1 (1970)
20. E.K. Storms, B.A. Mueller, J. Appl. Phys.
50
, 3691 (1979)
