
323
© Springer Nature Singapore Pte Ltd. 2017
P. Chandra et al. (eds.), Next Generation Point-of-care Biomedical Sensors
Technologies for Cancer Diagnosis, DOI10.1007/978-981-10-4726-8_14
X. Zhou (*) • T.I. Wong • J. Deng
Institute of Materials Research and Engineering, A*STAR (Agency for Science, Technology
and Research), 2 Fusionopolis Way, #08-03, Innovis, Singapore 138634, Singapore
e-mail: [email protected].edu.sg
L.L. Sun
Temasek Microelectronics Centre, School of Engineering, Temasek Polytechnic,
21 Tampines Avenue 1, Singapore 529757, Singapore
14
Point-of-Care Device withPlasmonic
Gold Nanoarray Sensing Chip
forBiomarker Detections
XiaodongZhou, TenItWong, LingLingSun, andJieDeng
14.1 Introduction
LSPR is generated on metal nanostructures upon the illumination of light whose
energy can be absorbed by the nanostructures and cause collective electron charge
oscillations on its surface. LSPR traps the light on the metal nanostructures surface
within tens of nanometers, where the light is strongly enhanced by tens to hundreds
of times. LSPR is particularly benecial for chemical or biological detections.
Because the sizes of the chemical or biological molecules are usually in a few nano-
meter range, they will be attached to the metal nanostructure surface roughly within
the coverage of the strong LSPR eld, thus the LSPR signal variation is relatively
specic to the molecule attachment, i.e., the molecules not bound to the metal nano-
structure will not much affect the detection signal, thus raw samples could possibly
be tested without wash or purications (Anker etal. 2008; Lee and El-Sayed 2006;
Sherry etal. 2005).
There are two methods to use LSPR for biosensors. One is to detect the shift of
the LSPR absorption due to the refractive index change upon molecule binding.
This is a direct assay that requires less time and cost; however, for some applica-
tions, the sensitivity might not be enough. The other is to utilize the LSPR to excite
the uorescent labels in a sandwich assay, because the labeled sandwich assay is
intrinsically more sensitive than direct assay, and LSPR eld is 10–100 times
stronger than the incident light, the LSPR amplies the uorescent labels and

324
achieves quite high sensitivity. Since both methods can be applied on the develop-
ment of a point-of-care device, the selection of detection methods is based on sen-
sitivity requirements for different applications. The development of a LSPR-based
point- of- care device has a ow chart illustrated in Fig. 14.1. The rst step is to
identify the nanoparticles and the related bioassay that can meet the requirement of
the specic application. After which, some plasmonic simulation is required to
identify the plasmonic peak and the LSPR eld distribution, thus the suitable light
source can be found and the size, shape, and inter-distance of the nanoparticles can
be optimized.
LSPR can be generated by metal nanoparticles in solution, or metal nanostruc-
tures (random or periodic nanoslits, nanobars, nanoholes, or nanopillars/islands) on
a substrate (Kuwata etal. 2003; Mayer and Hafner 2011; Murphy etal. 2005). We
established our point-of-care device with periodic gold nanoarray on glass sub-
strate, because the substrate-based LSPR chip can be washed with incorporated
microuidic channels to reduce nonspecic binding, the periodic nanostructures
have much narrower LSPR bandwidth that helps improving the detection sensitiv-
ity, and gold resists oxidation and extends the shelf-life of the fabricated sensing
chips. Furthermore, gold functions with different kinds of biomolecules and eases
the biofunctionalization of the nanostructures. However, periodic gold nanostruc-
tures are difcult to be fabricated, so we have developed a whole process of fabri-
cating the gold nanopillar or nanohole array on glass.
According to the simulated plasmonic eld as well as the simulated and charac-
terized LSPR wavelength of the nanochips (Ghaemi etal. 1998; Wu etal. 2012a;
Wu etal. 2012b), the bioassay is established on the top of the gold nanoarray, and a
microuidic channel with samples ow through the nanochip will be fabricated.
The microuidic chip can be actuated and detected by the automated point-of-care
device, and the point-of-care device will also quantify and report the concentration
of the biomarker in the specimen.
14.2 Nanochip Design, Fabrication, andCharacterizations
14.2.1 Design oftheGold Nanoarray
We rst designed the gold nanopillars and gold nanoholes with various pitches and
sizes, and the relation of the LSPR peak location to the size, pitch of the gold nano-
hole arrays are shown in Fig.14.2. The gold nanostructures can be designed by
plasmonic simulations according to the Maxwell equations, which are applicable
for the electromagnetic eld and transmission/refraction/absorption calculations for
metal nanoparticles larger than several nanometers. Commercial software available
Nanostructure
simulation
Nanochip
fabrication
Assay
decision
Microfluidic
chip
Biofunction
-alization
Point -of-care
development
Fig. 14.1 The development process of a LSPR-based point-of-care device
X. Zhou et al.
325
for such simulations includes COMSOL, CST Microwave, etc. For the ease of mass
productive nanofabrication of the sensing chips, we simulated the gold nanopillar
and nanohole arrays on glass substrate, by setting the frequency-dependent dielec-
tric constant of gold using the data from Palik handbook (Palik 1998), and the
refractive indices for quartz, air, and water were taken as 1.52, 1.0, and 1.33,
respectively.
0
1E6
R = 75
T
= 100
X
Y
α
β
water
gold
glass
α
β
water
gold
glass
Hole @ 540 nm
Max = 11.815
Pillar @ 540 nm
Max = 25
ab
cd
e
f
(75,100)
(60,150)
(100, 90)
=(R, T)
(50, 400)
(125, 90)
(150, 90)
(175, 90)
(190,90)
050 100 150 200
Intensity (W/m
2
)
x (nm)
80
70
60
50
40
30
20
10
0
400 500600 700800 900
Wavelength (nm)
gold thin film
sensor structure
optical power absorption [%]
660
650
640
630
620
610
100150 20
02
50
diameter (nm)
1.0
0.8
0.6
0.4
0.2
0.0
400 500600 700800 900100
0
Transmission
Absorption
Reflection
Wavelength (nm)
mode α wavelength (nm)
Power (W)
670
660
650
640
630
620
50 100 150 200 250 300
thickness (nm)
720
680
640
600
560
300 350 400 450 500
pitch (nm)
mode α wavelength (nm)mode α wavelength (nm)
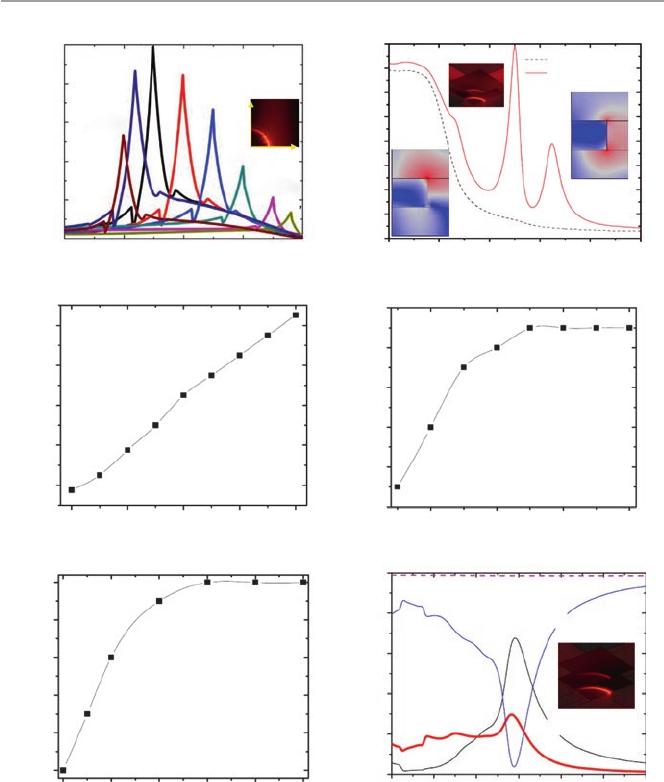
Fig. 14.2 Plasmonic peak and electromagnetic eld for water side light illumination. (a) is at
various gold nanohole dimensions with a xed pitch of 400 nm; (b) gold nanohole array
(p=400nm, R=75nm, T=100nm) showing the α and β modes; (c–e) the α peak shifts of gold
nanohole array in (b), when only varies the pitch, diameter, or gold thickness, respectively; (f) The
gold nanopillar array simulation with p=320nm, R=75nm, and T=50nm
14 Point-of-Care Device withPlasmonic Gold Nanoarray Sensing Chip

326
For biosensing, the gold nanoarray will be immersed in buffer with its reective
index similar to water. Plasmonics are generated at the top rims (at the water–gold
interface) and the bottom rims (at the gold–glass interface) of the gold nanostruc-
tures, and plasmonics on the top rims contributes more to the biosensing. The simu-
lated top rim plasmonic eld intensity is presented in Fig.14.2a for gold nanohole
arrays with a xed pitch of 400nm and various radii (R) and thicknesses (T). The
gold nanohole array with R=75nm and T=100nm yields the strongest plasmonic
eld of 9.538×10
5
W/m
2
(in our simulations, the incident power was 1nW within
a quarter of a single nanohole area, i.e., 200nm×200nm area). Figure14.2a also
indicates that the strongest plasmonic signal is at the rim of the gold nanoholes, and
this intensity drops to a half when X-axis distance is 6 nm away from the rim.
Figure14.2b further demonstrated that two plasmonic modes α (at around 647nm)
and β (at around 730nm) will be generated at the top and bottom rims of the gold
nanoholes, respectively. In comparison, in Fig.14.2b, 100nm gold lm on the glass
substrate presents no obvious plasmonic peaks.
The inuences of the pitch, diameter, and thickness to the wavelength of the α
mode in Fig.14.2c–e indicate that the pitch will inuence the absorption the most,
and 400nm is the most suitable pitch for having a plasmonic peak at around 647nm
which is suitable for red uorescent dye excitations. Figure 14.2c–e shows that
200nm of pitch variation (from 300 to 500nm) will cause a linear peak wavelength
shift of 180nm (from 555 to 735nm), 50nm of diameter variation (from 125 to
175nm) will cause a wavelength shift of 10nm (645–655nm), while 50 nm of
thickness variation (from 50 to 100nm) will cause a wavelength shift of 27nm
(from 620 to 647nm). It is also well known that engineering errors are unavoidable
in the gold nanostructure fabrications. So for different nanohole parameters, the
plasmonic peak absorption variation of the α mode can be roughly estimated by Δλ
=(∂λ/∂p)×Δp+(∂λ/∂D)×ΔD+(∂λ/∂T)×ΔT, with ∂λ/∂p=0.9, ∂λ/∂D=0.2 and
∂λ/∂T=0.54.
The gold nanopillar array with p=320nm, R=75nm, and T=50nm is also
used in our work, and its transmission, refraction, and absorption peaks in Fig.14.2f
present two plasmonic peaks at 540 and 690nm. For our work of using green light
to excite the quantum dot (QD) label in the sandwich assay, this kind of gold nano-
pillar array provides twice stronger plasmonic eld (by comparing the insets in
Fig.14.2b, f), thus outperforms the gold nanohole array of p=400nm, R=75nm,
and T=100nm.
14.2.2 Gold Nanoarray Fabrication
Based on our design, the gold nanohole array (with p = 400 nm, R = 75 nm,
T=100nm) and gold nanopillar array (with p=320nm, R=75nm, and T=50nm)
are our targets for nanofabrication. For point-of-care device, the gold nanoarray is
expected to be disposable, thus the mass fabrication of the gold nanochip is a key
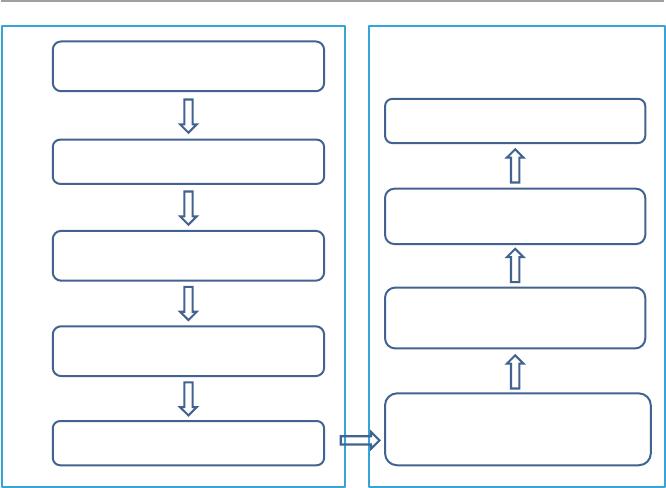
enabling technology for point-of-care devices. We have developed a mass fabrica-
tion method as illustrated in Fig.14.3. It includes two parts, the rst is the nickel
X. Zhou et al.
327
GOLD NANOPATTERN
MASS FABRICATION
Nano patterns design for electron
beam lithography.
Electron beam resist coating on
silicon substrate.
Electron beam lithography process
and development.
Metal seeding layer, coating on the
sample.
Nickel plating process and separation
of the nickel mold and substrate.
Photoresist coating on glass wafer
and use the sample together with
the nickel mold for nanoimprinting.
Nanoimprinting and follow by UV
curing inside UV oven.
Gold depostion and then the lift-off
process.
RIE to remove the residual of
photoresist.
M
O
U
L
D
F
A
B
R
I
C
A
T
I
O
N
Fig. 14.3 Mass fabrication of metal nanostructures on glass wafer. The left side indicates the
nickel mold fabrication process, while the right is the gold nanostructure mass fabrication
process
mold fabrication, and the second part the gold nanoarray mass fabrication via nano-
imprinting (Wong etal. 2013).
Electron-beam (e-beam) lithography is a technology of using focused e-beam to
expose the resist which is sensitive to the e-beam (Altissimo 2010; Chen 2015). The
e-beam lithography can reach a resolution of a few nanometers, and it is a revolu-
tionary nanotechnology for obtaining precise and controllable nanostructures in any
shape. E-beam lithography has been heavily applied in industry and nanotechnol-
ogy research, for fabricating the accurately designed nanopatterns in plasmonics,
semiconductors, surface property modied materials such as for anti-fouling or
anti-fogging, and photonic waveguides. Although nanoimprinting can replace the
e-beam lithography in some cases, its master mold is produced by e-beam
lithography.
In our work, the e-beam lithography is used to fabricate the nanochips in two
ways: the rst is the fabrication of the gold nanostructure array directly by e-beam
lithography (Song etal. 2015a), the other is to make a nickel mold which is used to
get the gold nanostructure array through nanoimprinting (Wong etal. 2013; Song
etal. 2015a; Song etal. 2015b). In either case, for reducing the cost of nanochip
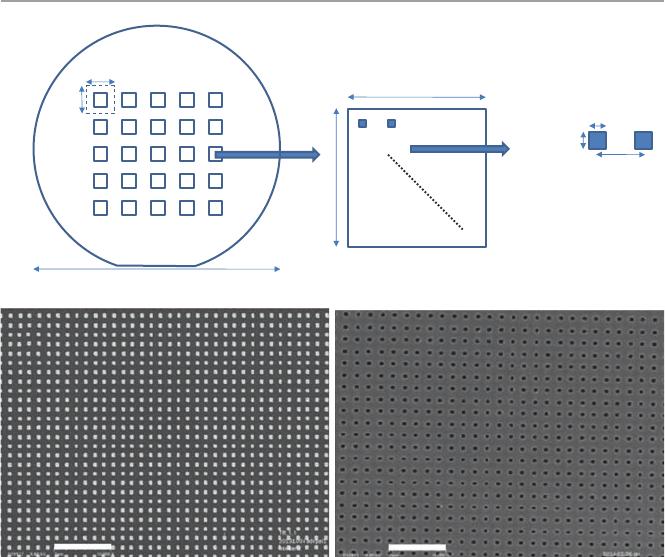
fabrication, e-beam lithography is conducted with 25 pieces of 1cm×1cm dies
arranged on one 4″ wafer. The chip arrangement is shown in Fig.14.4a. Each die
has its central 1.8mm×1.8mm area lled with fabricated 140nm×140nm sized
14 Point-of-Care Device withPlasmonic Gold Nanoarray Sensing Chip
328
nanohole or nanopillar array. We fabricated the nanoarray in square shape because
e-beam writes the squares slightly faster, and after nanofabrication, the gold
nanopatterns will have some round corners anyway.
The main caveat of the e-beam lithography, however, is its slow writing speed,
which substantially increases the cost. E-beam writing time includes the beam set-
tling time (i.e., the alignment time and beam shift time, as well as the time for beam
adjustment) and e-beam resist exposure time. According to the size of the nanopat-
terns, we optimized process and managed to reduce the e-beam resist exposure time
by using the writing voltage of 100keV, eld size of 300μm, lower dot map of 20k,
and a comparatively high current of 800pA on a single Gaussian beam (ELS-7000)
e-beam writing system. We did a series of experiments to compare, and found that
a minimum dose of 178μC/cm
2
for positive ZEP e-beam resist is required, in order
to get a reasonably high quality e-beam pattern for nanopatterns with 150nm size.
Compared with the normally used current of 200pA and dosage of 320μC/cm
2
for
e-beam writing on the ZEP, 800pA current and 178μC/cm
2
dose combination will
reduce the resist exposure time to 1/7 (Deng etal. 2016). This is very important,
because for the 4″ wafer e-beam writing shown in Fig.14.4a, even with our acceler-
ated e-beam writing recipe, it took 32h to complete. For the negative e-beam resist
NEB22, it can be exposed at 800pA current with 110μC/cm
2
dosage. Following the
140 nm
140 nm
Pitch = 400 nm for
nanohole array,
Pitch = 320 nm for
nanopillar array
1.8 mm
1.8 mm
………….
…………………
Diameter = 4 inch
1 cm
1 cm
a
b
c
Fig. 14.4 (a) The arrangement of nanochips on a 4″ wafer with 25 dies, and scanning electron
microscope (SEM) images of the fabricated gold (b) nanopillar and (c) nanohole arrays under
10,000× magnication, where the scale bar represents 2μm
X. Zhou et al.

329
e-beam exposure, the e-beam resist was developed for a few minutes to get the
nanopatterns.
In order to reduce the cost while keeping the high precision of the designed gold
nanohole array, nanoimprinting technology was adopted for the nanochip fabrica-
tion, because nanoimprinting can achieve sub 5nm resolution with high throughput
for nanolithography (Austin etal. 2004).
After the nanoarray pattern was written by e-beam lithography on silicon wafer,
the wafer was used for electroplating to get a nickel mold. To fabricate the nickel
mold, a thin metal seed layer was coated on the patterns to initiate the electroplating
process. Two-step plating was adopted: at the beginning 12μm of nickel was slowly
electroplated on the silicon wafer at a low current density of 0.7A/dm
2
, followed by
a high current density of 12A/dm
2
to electroplate up to 300μm of nickel. After
which, the nickel mold was separated from the silicon wafer. Trion plasma etching
system with O
2
gas was used to clean the nickel mold for 3min, and the silicon
wafer was recycled (Wong etal. 2013).
To nanoimprint, UV curable photoresist was selected, taking its advantage of using
a lower pressure (10bar) and lower temperature (as low as room temperature) for the
imprinting process compared with thermal nanoimprinting (30–40 bar, >100 °C),
which greatly reduces the rate of wafer breakage in nanoimprinting. The nickel mold
was rst coated with an anti-sticky layer of (Heptadecauoro-1,1,2,2-tetra hydro-
decyl) trichlorosilane (FDTS), then it was nanoimprinted on the UV curable photore-
sist coated on a 4″ glass wafer with the photoresist cured by UV light source.
After nanoimprinting, the nickel mold was easily demolded, and was cleaned for
reuse. Reuse of the nickel mold is critical, because the mold is generally fabricated
through e-beam lithography which is the major cost for the process to fabricate the
nanochips. The cost saving for fabricating the nanochip is tremendous if a nickel
mold can be reused, and nickel mode is reported to be durable for 10,000 times of
imprinting without obvious damage (Rai-Choudhury 1997).
The residue of the UV curable photoresist on the glass wafer was removed by
etching in an Oxford reactive ion etching system. The glass wafer was place in a
Dexton Explorer e-beam deposition machine, to rst deposit a few nanometer
(<5nm) of chromium to enhance the adhesion of the gold on glass, then deposit the
gold lm on the top of the chromium layer. Finally, the resist was lifted off at room
temperature to obtain the gold nanoarray.
The fabricated gold nanopillar or nanohole arrays on the 4″ glass wafer were diced
into 1cm×1cm chips by diamond blade using a Disco dicing saw. The fabricated pat-
terns were viewed under scanning electron microscope (SEM) or atomic- force micros-
copy (AFM) for checking their morphology and quality. The fabricated gold nanopillar
and nanohole arrays are presented in Fig.14.4b and Fig.14.4c, respectively.
14.3 Point-of-Care Device Based onLSPR Wavelength Shift
A point-of-care device using the LSPR wavelength shift is developed for the mea-
surement of cardiac troponin I (cTnI) in serum, as illustrated in Fig. 14.5. In
Fig.14.5a, white light transmitted through an optical ber bundle was collimated
14 Point-of-Care Device withPlasmonic Gold Nanoarray Sensing Chip
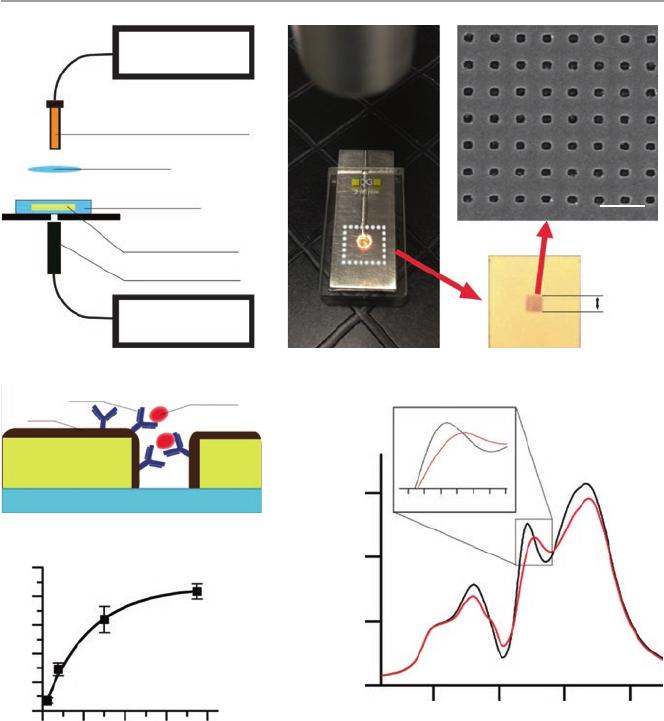
330
and illuminated on the top of the gold nanohole array (p=400nm, R=75nm, and
T=100nm) placed inside a transparent ow cell made of thermoplastic elastomer
(Fig.14.5b). The transmitted light spectrum was collected by another ber bundle
and recorded by a UV-vis spectrometer HR2000+ (Ocean optic, Dunedin, FL, USA)
in the range of 300–1000nm. Each frame of transmission was collected using an
acquisition time of 20ms, and the nal spectrum was produced by averaging 100
frames within 2s. Spectra were analyzed and evaluated using OriginPro 9, and the
transmission peaks were identied using a Lorentz-based method (Ding etal. 2015).
SAM
Antibody
Au
Glass
Analyte (cTnl)
Light source
Illumination fiber
Lens
Liquid cell
Sensor chip
Detection fiber
Spectrometer
15
12
9
6
3
0
020406080
Serum cTnl Concentration (ng/ml)
Peak Shift (nm)
a
1.5 mm
1.5 µm
b
15000
10000
5000
0
500 600 700 800
Wavelength (nm)
Transimission (arb. units)
644.07
654.85
620 630 640 650 660 670 680
Wavelength (nm)
d
e
c
Fig. 14.5 The point-of-care device based on the LSPR shift of a nanohole array (reproduced with
permission from reference Ding etal. (2015)). (a) Optical setup for the nanohole sensor detection.
(b) A liquid cell seals the gold nanohole array (with p=400nm, R=75nm, and T=100nm lling
a sensing area of 1.5mm×1.5mm) for the optical detection. (c) is the bioassay on the gold sur-
face, (d) shows the transmission spectrum of the nanochip upon cTnI (at 30ng/mL concentration)
binding, and (e) is the characterization curve of the detected cTnI concentration versus the LSPR
peak shift
X. Zhou et al.

331
The direct assay in Fig.14.5c was established by rst forming an amine-reactive
self-assembly monolayer (SAM) incubating the nanochips in ethanolic solution of
0.4mM of 10-carboxy-1-decanethiol and 1.6mM of 1-octanethiol for 12h at room
temperature, then washed thoroughly with pure ethanol and dried in room tempera-
ture. Then the chip was incubated in a mixture of sulfo-N-hydroxysuccinimide
(sulfo-NHS) and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) (Bio-
Rad, Hercules, CA, USA) to activate the carboxylic group of the SAM for 15min.
Next, 50μL of 200μg/mL anti-troponin antibody 560 (Hytest, Finland) in acetate
buffer at pH4.5 was spotted on the chip surface and incubated for 30min. Finally,
the sensor chip was immersed in 1 M of ethanolamine-HCl solution (Bio-Rad,
Hercules, CA, USA) for 15min to deactivate the unreacted esters, followed by a
rinse with deionized water and dried with nitrogen gas at room temperature. The
sensing chip is blocked by 1% bovine serum albumin (BSA) solution for 15min to
prevent the nonspecic binding, then 50 μL of human cTnI standard in serum
(Phoenix Pharmaceuticals, CA, USA) in the concentrations of 75, 30, 7.5, or 2.5ng/
mL was applied and incubated for 30min. The sensor chip was rinsed with PBS
solution three times after incubation, and inserted into the measurement cell con-
taining PBS buffer to record its transmission spectrum.
The binding of the cTnI on anti-troponin antibody on the sensor chip will shift
the plasmonic peak at the wavelength of 644.07–654.85nm (Fig.14.5d). The peak
at 644.07nm is the α peak in Fig.14.2b which has the plasmonic eld concentrated
on the gold–water interface where the bioassay is anchored. Thus the LSPR bio-
sensing for this plasmonic peak monitoring is relatively sensitive. Figure 14.5e
shows the characterization curve for cTnI detection, with error bars obtained by
measuring the same troponin concentrations with three individual nanochips.
The performance of this point-of-care device is found to have a full half- magnitude
width (FHMW) of 32.84nm, gure of merit (FOM) of 10.5, σ of signal at 0.017nm,
and signal-to-noise ratio equals to 256 (at 7.5ng/mL troponin in serum), and the limit
of detection (LOD) based on three times of σ is identied to be 0.55ng/mL.
14.4 Point-of-Care Device Based onFluorescent Label
Enhancement
We have developed several sandwich bioassays based on plasmonic-enhanced uo-
rescent labels to respectively detect the prostate specic antigen (PSA) for prostate
cancer, thrombin for blood clots, and procalcitonin (PCT) for sepsis. When estab-
lishing such a bioassay, a few factors are considered. The rst is that the distance of
the uorescent label should be around 10–20nm to the gold nanostructure surface,
so that the dyes are enhanced by the plasmons instead of being quenched by the
strong plasmons. The second is that the excitation wavelength of the dye must be
close or slightly shorter than the LSPR generated by the gold nanostructures, so as
to make sure that the light being used to excite the uorescent dyes are amplied by
the plasmon. Here we use quantum dot (QD) as labels, because compared with
organic uorescent dyes, the QDs are about 20 times brighter, and they have a
broadband of excitation wavelength range (Song etal. 2015a).
14 Point-of-Care Device withPlasmonic Gold Nanoarray Sensing Chip

332
These three kinds of biomarkers are all detected with a sandwich assay on the
gold nanoarray surface using the QD as uorescent label. However, they are still
slightly different. The rst type of assay demonstrated for PSA detection is to use
the tris(2-carboxyethyl) phosphine (TCEP) solution to cleave the PSA monoclone
capturing antibody (cAb) into a half and utilize the –SH link to bind the PSA captur-
ing antibody, which is an effective way to reduce the distance between the QD label
and the gold surface (Song etal. 2015a). The second is to use aptamer to capture the
thrombin, because compared with capturing antibodies, aptamers are relatively
cost-effective and with high repeatability, durability, reproducibility, and it is rela-
tively easy for the surface regeneration when the nanochip is planned to be reused
(Song etal. 2015b). The third is to use the whole PCT capturing antibody to capture
the PCT on the gold surface (Sun etal. 2016). All these three kinds of bioassays can
be detected by a microscope or a camera, and further the point-of-care device can
be fabricated based on imaging of the uorescent labels.
14.4.1 Bioassay forPSA Detection withCleft Antibody
PSA is a US FDA approved cancer biomarker indicating the risk and existence of
prostate cancer for males, with a cutoff value of 4ng/mL for prostate cancer screen-
ing. Since the antibody is about 14nm high, a standard sandwich assay will make
the QD labels probably more than 30nm above the gold nanostructure surface and
hinder the enhancement by the plasmons. In order to further reduce the distance
between the QDs and the gold surface, in our method, the whole capturing anti-
body was cleft into a half by TCEP solution (Song etal. 2014). This was achieved
by adding 5μL of TCEP into 50μL of 250μM cAb in PBS (1.26mg/mL, 8.4μM).
After incubating at room temperature for 30 min to allow the mixtures to fully
react, G25 column was used to purify the cleft cAb. The cleft cAb solution was
stored at 4°C, and diluted to 50μg/mL in 4-(2-hydroxyethyl)-1-piperazineethane-
sulfonic acid (HEPES) buffer (prepared by mixing 10mM HEPES with 150mM
NaCl) before use.
Compared with the whole capturing antibody, the cleft antibody is about half of
its size, and since the –SH link will be attached to the gold surface, the cleft cAb are
with their fragment antigen-binding sites facing upwards on the gold surface. The
cleft cAbs in an orientation controlled state have more binding sites compared with
the randomly orientated whole cAb, which will further increase the sensitivity for
biosensing. With the cleft cAb, a sandwich assay of “cleft cAb– PSA– biotinylated
dAb– QD with streptavidin” was immobilized on the gold surface (Fig.14.6a), and
the distance of the gold nanostructure to the QD is about 20–25nm, which is within
the distance for the QDs to be effectively excited by plasmons (Song etal. 2015a).
The QD-655 has an emission peak at the wavelength of 655nm. Practically, we
used green light at the wavelength of 540nm to illuminate the chip from the bioas-
say side and detect the QD emission from the same side of the chip. As the light
sheds from the top of the chip, the plasmonic elds for the gold nanohole array
(p=400nm, R=75nm, and T=100nm) and gold nanopillar array (p=320nm,
X. Zhou et al.
