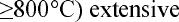
Void Morphology In NiAl
M. Zakaria and P.R. Munroe
Electron Microscope Unit
University of New South Wales
Sydney NSW 2052, Australia
ABSTRACT
Void formation in stoichiometric NiAl was studied through controlled heat treatments and
transmission electron microscopy. Voids formed at temperatures as low as 400°C, but dissolved
during annealing at 900°C. Both cuboidal and rhombic dodecahedral voids were observed, often
at the same annealing temperature. At higher annealing temperatures (
dislocation climb was noted. The relative incidence of void formation and dislocation climb can
be related to the mobility of vacancies at each annealing temperature. Further, differences in void
shape can be described in terms of their relative surface energy and mode of nucleation.
INTRODUCTION
Large supersaturations of vacancies can be quenched into NiAl following high temperature
(>1000°C) heat treatment. During subsequent annealing at lower temperatures the excess
vacancies are removed from the lattice through either dislocation climb or void formation. A
number of workers have studied voids in NiAl, but the behaviour of these defects has not been
unambiguously defined [1-5]. That is, the conditions under which voids form, their shape, size
and range of stability have not been clearly defined. Two distinct void shapes, cuboidal and
rhombic dodecahedral, are observed, but their incidence cannot be unambiguously related to
particular heat treatment conditions or alloy composition. In some cases, dislocation loops, rather
than voids, are formed [6-8]. We have examined the structure of voids in stoichiometric NiAl as
a function of annealing temperature over a range from 400°C to 900°C. Detailed descriptions of
the observed microstructures can be found elsewhere [9]. In this paper experimental observations
will be summarised more briefly, the principal aim of this paper is to discuss the relative
incidence of void formation and dislocation climb, and the variations in void shape.
EXPERIMENTAL METHODS
Nominally stoichiometric NiAl was prepared by arc melting under an argon atmosphere. The
material was remelted several times to improve homogeneity. Chemical analysis indicated that
composition was close to stoichiometry. Samples were annealed, in air, at 1300°C for 2 hours
and cooled to room temperature by air-cooling. Subsequent annealing was performed at
temperatures ranging from 400°C to 900°C for 1, 5 or 24 hours at each temperature. Thin foils
for transmission electron microscopy (TEM) study were prepared and examined in a JEOL
2000FX TEM operating at 200kV.
Mat. Res. Soc. Symp. Vol. 646 © 2001 Materials Research Society
N5.6.1

RESULTS AND DISCUSSION
The microstructure of NiAl prior to annealing was examined. As expected, the microstructure
was single phase with equaixed grains about 200µm in diameter. Following homogenization at
1300°C the microstructure contained fine (~ 20nm diameter) dislocation loops, with <001>
Burgers vectors. A very small number of fine (~20nm diameter) voids were also noted. These
voids were presumably formed during cooling, where there is presumably sufficient time for
some vacancy agglomeration to occur.
Following annealing at 400°C for times up to 5 hours, the defect structures were broadly
similar to the specimen air-cooled from 1300°C. However, following annealing for 24 hours at
400°C a number of small (~10nm) cuboidal voids was observed (figure 1). In contrast, other
workers have observed dislocation loops in single crystal Ni-53Al annealed at 425°C for 1 hour
after prior air-cooling from 1175°C. However, Eibner et al. [5] also observed cuboidal voids
(~10nm in diameter) following annealing at 400°C for 32 hours following water quenching from
1600°C. On the other hand, Epperson et al. [3] observed rhombic dodecahedral voids in single
crystal Ni-50.4Al annealed at 400°C for 22 hours following water-quenching from 1600°C.
Figure 1. Bright field TEM image of NiAl following annealing at 1300°C and subsequent annealing at 400°C
for 24 hours. Very fine (10nm diameter) voids can be observed.
Following annealing at 500°C for 1 hour, cuboidal voids, ~40nm in diameter, were noted
(figure 2a). A few larger voids (~100 nm) were also observed (marked 'v'), possibly associated
with growth of pre-existing voids formed during initial heat treatment. In contrast, after
annealing for either 5 or 24 hours rhombic dodecahedral voids were seen, typically ~50-100 nm
in diameter (figure 2b). Yang and Dodd did not observe voids in NiAl following annealing at
500°C [2], but others have noted rhombic dodecahedral voids after quenching from high
temperature and annealing at 500°C [1,4]. In contrast, Ball and Smallman [6] observed
dislocation loops in near-stoichiometric NiAl containing 0.06%C annealed at 500°C for 15
minutes after quenching from 1300°C.
After exposure for 1 hour at 600°C rod-shaped voids with lengths parallel to {001} were
observed (figure 3a). These voids were between 600 and 1200 nm in length and ~40 nm in
width. However, following annealing for 5 hours only rhombic dodecahedral voids were
observed. The void diameter was about 50-100nm, and some void coalescence was noted (figure
3b). The elongated voids were not observed. These elongated voids were very similar to those
N5.6.2

observed by Yang and Dodd [2] in near-stoichiometric NiAl containing 0.05%C following the
same heat treatment conditions. However, it is not clear why these voids are not present after
annealing for longer times. The void size, shape and density following annealing at 600°C for 5
hours is also similar to that noted by Yang and Dodd [2] and Epperson et al. [3].
Figure 2. Bright field TEM images of NiAl following annealing at 1300°C and subsequent annealing at 500°C
for a) 1 hour and b) 5 hours.
Figure 3. Bright field TEM images of NiAl following annealing at 1300°C and subsequent annealing at 600°C
for a) 1 hour and b) 5 hours
Following annealing at 700°C for 1 hour, cuboidal voids, with diameters between 50 and
100nm, were observed (figure 4a) A similar defect structure was noted after 5 hours. This is
consistent with the observations of Eiber et al. [5]. However, following annealing for 24 hours
rhombic dodecahedral voids were noted, although in some regions of this specimen a higher
dislocation density was noted (figure 4b). The dislocations exhibited a <001> Burgers vector and
were edge in character. Numerous jogs were observed along their line length. Often these
dislocations were noted to interact with any voids present (see region marked V).
Heat treatment at 800°C lead to the formation of very large (100-300nm) cuboidal voids
following annealing for 24 hours (figure 5). In contrast, Yang and Dodd [2] observed larger
(~160nm diameter) rhombic dodecahedral voids in near-stoichiometric alloys containing 0.05%C
following annealing at 800°C. Whilst other workers observed only dislocation loops in near-
stoichiometric NiAl annealed at 800°C for 30 minutes [1].
Y
Y
Y
a)
a)
b)
b)
N5.6.3

Figure 4. Bright field TEM images of NiAl following annealing at 1300°C and subsequent annealing at 700°C
for a) 1 hour, and b) 24 hours
Figure 5. Bright field TEM images of NiAl following Figure 6. Bright field TEM images of NiAl following
annealing at 1300°C and subsequent annealing annealing at 1300°C and subsequent annealing
at 800°C for 24 hours. at 900°C for 24 hours.
At 900°C the void density was noted to be lower than at lower annealing temperatures.
Further, the void density was noted to decrease with annealing time. Dislocation loops, which
exhibited jogs along their length, were more commonly noted in specimens annealed at this
temperature (figure 6).
It is clear that variable void formation (size, shape and density) took place at different
annealing temperatures, or even at the same temperature for different annealing times. Both
rhombic dodecahedral and cuboidal voids were observed. It was also observed that void
shrinkage occurred at 900°C, while at lower to intermediate temperatures (400-700°C), void
nucleation and growth occurred. In spite of the large variations in void shape and size noted,
these observations are broadly consistent with those of other workers [1-5].
The equilibrium thermal vacancy concentrations at the annealing temperatures used can be
calculated (assuming an energy of formation, E
f
, of 1.45eV [4]), these are presented in Table 1.
Clearly, a high concentration of vacancies form during heat treatment at 1300°C. The large
density of voids subsequently formed indicates that a significant fraction of these were retained
on air-cooling. The excess vacancies were removed during subsequent annealing by either void
nucleation and growth, or dislocation climb. Vacancies can also be removed by migration to
Y
a)
Y
b)
N5.6.4

vacancy sinks such as grain boundaries, although the large grain size in these alloys would
mitigate against this.
Table 1. Vacancy concentration (%) and number of vacancy jumps, J
v
, at different annealing temperatures, and
for different annealing times
Temperature Vacancy jumps, J
v
(°C)
Vacancy
concentration
n/N (%)
1 hr 5 hr 24 hr
400 7.92x10
-4
3.94x10
4
1.97 x10
5
9.47 x10
5
500 3.96 x10
-3
6.64 x10
6
3.32 x10
7
1.59 x10
8
600 6.84 x10
-3
3.45 x10
8
1.73 x10
9
8.29 x10
9
700 0.0183 7.97 x10
9
3.99 x10
10
1.91 x10
11
800 0.0408 1.03 x10
11
5.13 x10
11
2.46 x10
12
900 0.0795 8.53 x10
11
4.27 x10
12
2.05 x10
13
1300 0.49 - - -
The vacancy migration energy, E
m
has been estimated for NiAl to be about 2.3eV [4]. On this
basis the number of jumps, J
v
for each annealing condition was calculated and the results are also
summarised in Table 1. On the basis of a random walk process, and assuming that <100> jumps
take place, the vacancy migration distance can be calculated. For 1 hour at 400°C, a vacancy will
move ~50 nm, but at 900°C for 24 hours a vacancy can migrate about 1mm. The high density of
voids observed at lower annealing temperatures presumably corresponds to the short distances
over which the vacancies were able to move; that is, they cluster to form voids. However, at
higher temperatures, vacancies migrate much further, thus they would be able to diffuse to
dislocations, where they can be annihilated by climb, or they migrate to grain boundaries.
The highest void densities were noted at low or intermediate temperatures (
°C), where
the equilibrium thermal vacancy concentration is relatively low. Thus, the driving force for
vacancy removal is high, but vacancy mobility is low. A large number of vacancies may be
driven out of supersaturation and with their limited mobility may cluster locally to form a high
density of voids. At higher annealing temperatures (
°C) lower void densities were noted.
Here, the equilibrium thermal vacancy concentration is much higher, so the driving force for
vacancy removal decreases. These vacancies are much more mobile and may diffuse to grain
boundaries or to dislocations.
At 900°C voids dissolved as annealing time increased. This is perhaps related to both
mechanisms of vacancy removal being in operation together. That is, voids form initially at this
temperature, but as annealing time increases more vacancies migrate to dislocations where they
are annihilated. This lowers the retained vacancy concentration so voids may dissolve and go
back into solution to maintain the equilibrium vacancy concentration at this temperature.
Both rhombic dodecahedral and cuboidal voids were observed in this study. Often, different
void shapes were observed at different times at the same annealing temperature. The origins of
these different shapes are unclear. It is possible that void shape is affected by both ease of
nucleation and surface energy effects. Turning firstly to surface energy effects, cuboidal voids,
with faces parallel to {001}, will have a different surface energy to that of rhombic dodecahedral
voids with most faces parallel to {011}. Clapp et al. [10] estimated that for NiAl the surface
energy of {001} was ~1 J/m
2
, while the surface energy of {011} was ~1.5 J/m
2
. If the void
diameter is taken nominally as 50 nm then for cuboidal voids, there will be six faces, each of
N5.6.5
which has an area of (50x50)10
-9
m
2
. Using the model for shape of the rhombic dodecahedral
voids suggested by Epperson et al. [3], there will be six {011} “prism” faces with an area of
(50x50)10
-9
m
2
plus two {001} “basal” faces with an area of 7.5x10
-15
m
2
. Thus, a cuboidal void
has an energy of 1.5x10
-14
J
and a rhombic dodecahedral void an energy of 3x10
-14
J. On this
basis, cuboidal voids should be energetically more stable. It is possible that rhombic
dodecahedral voids exist due to the differing nucleation mechanisms that may operate.
The B2 structure of NiAl consists of two interpenetrating simple cubic cells, where Al atoms
occupy one sublattice and Ni atoms the other sublattice. If two vacancies substitute on to nearest-
neighbour positions, this may lead to a plane of vacancies on {011}. Vacancy agglomeration on
these planes may then ultimately result in the nucleation and growth of rhombic dodecahedral
voids. Alternatively, if vacancies substitute on to next nearest-neighbour positions, that is two
vacancies on the same sublattice, a plane of vacancies on {001} is more likely to result and this
may then lead to the nucleation and growth of cuboidal voids. It would appear, therefore, that
both mechanisms occur here. This is consistent with the work of Fu et al. who suggested that
thermal vacancies in NiAl do not exhibit a preference for any specific lattice site [11].
CONCLUSIONS
Vacancy formation has been studied in stoichiometric NiAl, heat treated to produce a
supersaturation of thermal vacancies, over a temperature range from 400°C to 900°C. Both
cuboidal and rhombic dodecahedral shaped voids were noted, often both void types were noted
at a single annealing temperature. At lower annealing temperatures void formation was the
preferred method of removal of thermal vacancies, but at higher temperatures vacancies were
more likely to be removed by dislocation climb. The shapes of the two vacancy types observed
were rationalized in terms of their relative surface energy and possible methods of nucleation.
REFERENCES
1. W. Yang, R.A. Dodd and P.R. Strutt, Metall. Trans. 3A, 2049 (1972).
2. W. Yang and R.A. Dodd, Scripta. Metall. 8, 237 (1974).
3. J.E. Epperson, K.W. Gerstenberg, D. Berner, G. Kostroz and C. Ortiz, Phil. Mag. A 38,
529 (1978).
4. A. Parthasarathi and H.L. Fraser, Phil. Mag. A 50, 89 (1984).
5. J.E. Eibner, H.J. Engell, H. Schultz, H. Jacobi and G. Schlatte, G, Phil. Mag. 31, 739
(1975).
6. A. Ball and R.E. Smallman, Acta. Metall. 14, 1517 (1966).
7. G.W. Marshall and J.O Brittain, Metall. Trans. 7A, 1013 (1976).
8. T.C. Tisone, G.W. Marshall and J.O Brittain, J. Applied Phys. 39, 3714 (1968).
9. M. Zakaria and P.R. Munroe, J. Mater. Sci., in submission (2000).
10. P.C. Clapp, M.J. Rubins, S. Charpenay, J.A. Rifkin and Z.Z. Yu, in High Temperature
Ordered Intermetallic Alloys III, edited by C.T. Liu, A.I. Taub, N.S. Stoloff and C.C.
Koch, (Mater. Res. Soc. Proc. 133, Pittsburgh PA, 1989) pp. 29-35.
11. C.L. Fu, Y.Y.Ye and M.H. Yoo, in High Temperature Ordered Intermetallic Alloys V
edited by I. Baker, R. Darolia, J.D. Whittenberger and M.H. Yoo, (Mater. Res. Soc. Proc.
288, Pittsburgh PA, 1993) pp. 21-32.
N5.6.6
