
Developing Criteria and Associated
Instructions for Consistent and Useful Quality Improvement
Study Data Extraction for Health Systems
Adrian V. Hernandez, MD, PhD
1,2
, Yuani M. Roman, MD, MPH
1,2
, and C. Michael White,
Pharm. D., FCP, FCCP
1,2,3
1
School of Pharmacy, University of Connecticut Evidence-based Practice Center, Storrs, CT, USA;
2
Department of Pharmacy, Hartford Hospital,
Hartford, CT, USA;
3
University of Connecticut School of Pharmacy, Storrs, CT, USA.
BACKGROUND: The Agency for Healthcare Research and
Quality (AHRQ) could devote resources to collate and as-
sess quality improvement studies to support learning
health systems (LHS) but there is no reliable data on the
consistency of data extraction for important criteria.
METHODS: We identified quality improvement studies
and evaluated the consistency of data extraction from
two experienced independent reviewers at three time
points: baseline, first revision (where explicit instructions
for each criterion were created), and final revision (where
the instructions were revised). Six investigators looked at
the data extracted by the two systematic reviewers and
determined the extent of similarity on a scale of 0 to 10
(where 0 represented no similarity and 10 perfect similar-
ity). There were 42 assessments for baseline, 42 assess-
ments for the first revision, and 42 assessments for the
final revision. We asked two LHS participants to assess
the relative value of our criteria.
RESULTS: The consistency of extraction improved from
1.17 ± 1.85 at baseline to 6.07 ± 2.76 after revision 1 (P <
0.001) and to 6.81 ± 1.94 out of 10 for the final revision (P
< 0.001). However, the final revision was not significantly
improved over the first revision (P = 0.14). One key infor-
mant rated the difficulty in finding and using quality im-
provement studies a 6 (moderately difficult) while the oth-
er a 4 (moderately difficult). When asked how valuable it
would be if AHRQ found and collated the demographic
information about the health systems and the interven-
tions used in published quality improvement studies,
they rated it a 9 (highly valuable) and a 6 (moderately
valuable).
CONCLUSION: Creating explicit instructions for extract-
ing data for quality improvement studies helps enhance
the consistency of data extraction. This is important be-
cause it is difficult for LHS to vet these quality improve-
ment studies on their own and they would value AHRQ’s
support in that regard.
KEY WORDS: quality improvement; data extraction; learnin g health
system.
J Gen Intern Med
DOI: 10.1007/s11606-020-06098-1
© Society of General Internal Medicine (This is a U.S. government work and
not under copyright protection in the U.S.; foreign copyright protection
may apply) 2020
INTRODUCTION
Quality patient care is an important part of health system accred-
itation and is increasingly tied to revenue through the hospital
value-based purchasing program.
1–3
In 2020, 55% of health
systems will see added net revenue from hitting performance
metrics while others will receive a financial penalty.
2
In its report,
Best Care at Lower Cost: The Path to Continuously Learning
Health Care in America, the Health and Medicine Division of the
National Academy of Sciences proposed the concept of the
learning health system to improve healthcare quality and out-
comes.
4
Regular health systems become learning health systems
when they internally identify areas of quality weakness and strive
to utilize the best knowledge to improve them.
5, 6
The Agency for Healthcare Research and Quality (AHRQ)
created a working group in 2017 that interviewed nine leaders
in quality and safety improvement in health systems to explore
how they used evidence.
7
Health systems looked to other
institutions within their buying group, institutions in a formal
consortium, or institutions reporting their experiences in the
biomedical literature, for quality improvement approaches.
However, finding the full spectrum of quality improvement
projects completed in an area from the medical literature is
difficult. There are many ways to improve the quality of care
including purchasing new equipment or software, dedicating
staff to champion changes, educational programs, email
reminders, electronic health record reminders, internal proto-
cols and guidelines, prior authorization, and limiting utiliza-
tion to a specialist or specialty service. Faced with multiple
approaches to improve, health systems need to identify
approaches that overlap with their strengths and are realistic
with their fiscal and staffing reality. The best approach in one
health system may not translate well to another and, in some
cases, may not even be feasible.
The AHRQ Evidence-based Practice Center (EPC) program
created a report on closing the quality gap in asthma care.
8, 9
Electronic supplementary material The online version of this article
(https://doi.org/10.1007/s11606-020-06098-1)containssupplementary
material, which is available to authorized users.
Received August 26, 2019
Revised March 13, 2020
Accepted July 30, 2020

The EPC program consists of institutions in the USA and
Canada that review relevant scientific literature on a wide
spectrum of clinical and health services topics to produce
various types of evidence reports. These reports may be used
for informing and developing coverage deci sions, quality
measures, educational materials and tools, clinical practice
guidelines, and research agendas.
8
In this asthma report, they
had to screen 3843 titles and abstracts to find the 171 relevant
quality improvement projects that had been conducted. The
interventions included self-monitoring, patient or caregiver
education, provider education, organizational change, auditing
of records and fee dback, pro vider remin ders, pati ent
reminders, and financial or nonfinancial incentives.
9
The inter-
ventions spanned from 1 to 60 months and some were in
outpatient primary care clinics, pulmonary clinics, home ,
school, community centers, and simultaneously applied mul-
tiple settings. The interventions were devised or provided by
different people including physicians, nurses, pharmacists,
health educators, and healthcare teams. In this example of a
single quality improvement systematic review, the effort need-
ed to find relevant data was immense as was the heterogeneity
of the intervention types, where the interventions took place,
and who provided or championed the interventions. These
factors make it difficult for individual health systems to iden-
tify the types of quality improvement studies that would be
especially relevant to them.
9
EPCs also conduct research on the methodology of evi-
dence synthesis and tagged the quality improvement literature
as needed additional assessment and understanding. In 2018, a
multi-EPC and AHRQ working group sought to develop a
process to assess and present useful information from quality
improvement projects to learning health systems.
8
There was
progress in defining quality improvement studies and gener-
ating a list of 33 candidate criteria (Table 1) describing fea-
tures of the interventions and the environment in which they
were conducted. Unfortunately, there was little similarity to
what was bei ng ex tracted by exper ienced system atic
reviewers, which was an impediment to progress, and work-
group activities were postponed. For example, for the criteria
entitled “Who is receiving the intervention?” had one data
extractor saying “Nurses” as compared with another that said
“Advanced Practice Nurses in the Surgical Intensive Care
Unit” or for the criteria “Duration of the intervention” had
one data extractor stating the duration was the extra time
needed to clean the patient’s room (e.g., 15 min) while another
stated it was the entire time the new protocol had been in place
(e.g., several months). Furthermore, these criteria were not
assessed by learning health system participants to identify
the ones of greatest value. Without this input, it is unclear
whether having 33 criteria overwhelms the reader without
providing value or the relative value of each of the criteria.
The University of Connecticut (UConn) EPC sought to
develop data extraction instructions for each criterion that
can enhance the consistency of data extraction among different
systematic reviewers and assess the difficulty health systems
have with identifying and using quality improvement studies.
METHODS
Assessing the Consistency of Data Extraction
We utilized studies contained in the AHRQ report entitled
“Closing The Quality Gap: A Critical Analysis of Quality
Improvement Strategies: Volume 5—Asthma Care” as our
data sources.
9
We chose this report because it was an extensive
review of quality improvement projects that had been con-
ducted by an EPC and the use of our criteria is envisioned to
result in enhancements to subsequent EPC reports like this one
should it turn out to be valuable. In the first phase of the study,
we used the criteria (Table 1) that the 2018 AHRQ working
group identified as potentially valuable for health systems to
understand how to use and implement QI studies.
8
These
criteria were selected from previous work in the quality im-
provement and implementation literature.
10–13
Our methodology is displayed visually in Figure 1.At
baseline, two experienced EPC systematic reviewers (AVH
and YMR, named authors in this article) extracted data for all
33 criteria from two different studies (Kamps et al. 2003 and
2004, and Brown 2004 ).
9
There was only a rudimentary
description of each criterion at this stage. When examining
Table 1 Criteria to Assess Quality Improvement Studies
1. Who is delivering the intervention
(e.g., provider types)?
10. Duration of the effect of the
intervention
19. Leadership commitment and
involvement
28. Population needs/
burden of illness
2. Who is receiving the intervention
(e.g., patient types)
11. Team composition (people
delivering the intervention)
20. Clinical champion
involvement
29. Geographic location
3. Provider demographics 12. External policies and incentives
required
21. Physical environmental
changes required
30. External factors
4. Recipient demographics 13. Required skills/training 22. Incentives 31. Organizational history
of change
5. Active vs. passive components 14. Number and description of
components
23. Implementation strategies 32. Fidelity
6. Discretionary vs. mandatory
components
15. A priori components vs. added
later/final
24. Types of intervention effects 33. Intervention adaptation
7. Duration of the intervention 16. Theoretical foundation 25. Organizational setting
8. Frequency of intervention 17. Which interventions are
independent
26. Financial setting
9. Intensity of the intervention 18. Cost of implementation 27. Organizational receptivity/
readiness
Hernandez et al.: Criteria and Instructions for Consistent QI Extractions JGIM
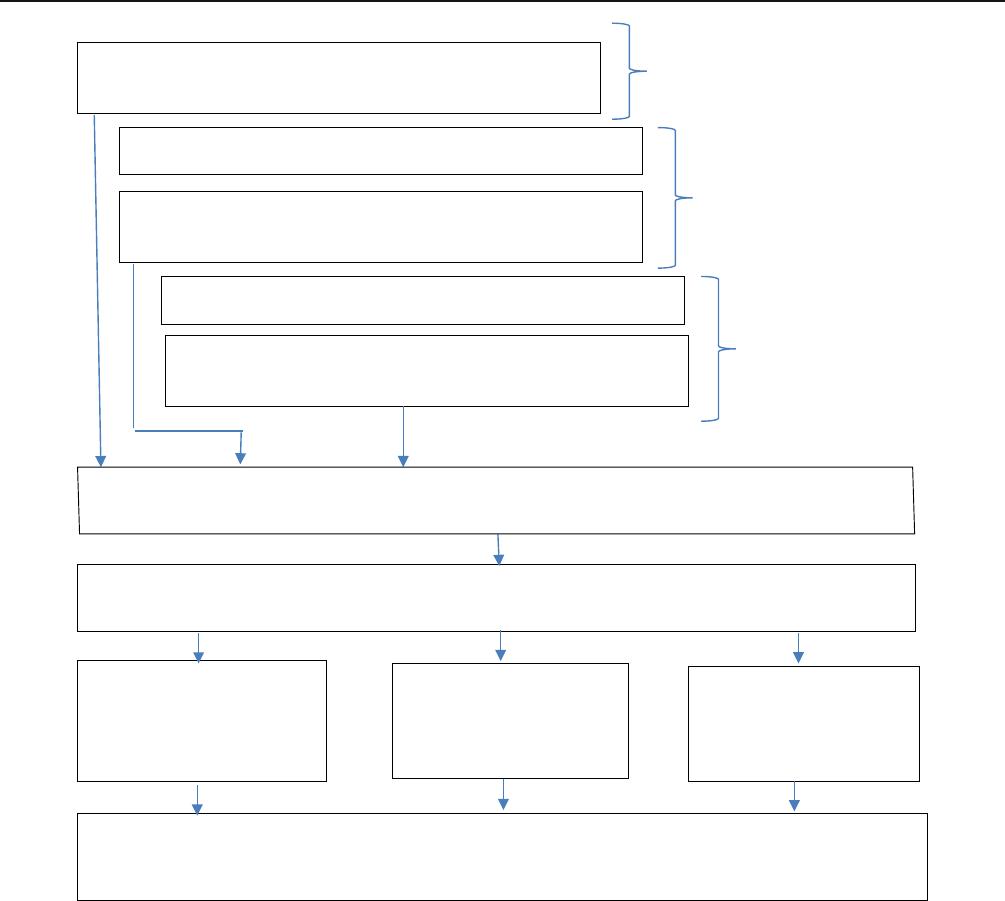
the consistency of the results, three investigators mutually
developed detailed instructions on how to extract study data
for each criterion. Two systematic reviewers then indepen-
dently extracted data for all 33 criteria from two other source
studies utilizing these instructions (first revision). Reviewing
inconsistencies among extractors led to research team to refine
the instructions for each criterion and reduce ambiguity. Using
the refined instructions (final revision), data was independent-
ly extracted by two systematic reviewers for each of the 33
criteria from two other studies. For example, criterion #7
Duration of Intervention was confusing at baseline as it was
not clearly described the different between duration of inter-
vention and length of intervention. After the first revision, we
added the following extra text: “Duration of intervention
should specify total hours/days of intervention. It comes from
the multiplication of frequency and intensity. Length of inter-
vention is the calendar time t he intervention ha s been in
place.” During the final revision, criterion #7 was not a source
of inconsistency anymore.
To assess the impact of the instructions on the consistency
of data extraction, an investigator randomly selected dually
extracted data from 7 of the 33 specific criteria (termed a data
field) listed in the Appendix at each phase using a random
permutations generator (Randomization.com – Third Genera-
tor). In all, seven dually extracted data fields were selected at
baseline, seven after the first revision of the instructions were
completed (first revision), and seven after the final instructions
were completed (final revision). Six investigators looked at the
data extracted by one of the systematic reviewers and then the
other for the same field and determined the extent of similarity
on a scale of 0 to 10 (where 0 represented no similarity and 10
perfect similarity of extraction). This meant there were 126
assessments of the similarity of extracted data (42 assessments
for baseline, 42 assessments for first revision, and 42
Two Systematic Reviewers Independently Extracted
Data from 2 Studies on 33 Criteria
Two Systematic Reviewers Independently Extracted
Data from 2 Studies on 33 Criteria at Baseline
Guidance on How to Extract Data Revised
Guidance on How to Extract Data Revised
BASELINE
Two Systematic Reviewers Independently Extracted
Data from 2 Studies on 33 Criteria at Baseline
FIRST
REVISION
FINAL
REVISION
TWO
Seven of 33 Criteria Randomly Selected Per Phase (BASELINE. FIRST REVISON,
FINAL REVISION) and Dually Extracted Data from Both Systematic Reviewers Used
Six Investigators Evaluated Each Set of Dually Extracted Data for Each Criteria Selected
and Determined How Similar They Were on a Scale of 0 to 10
BASELINE
42 Consistency Ratings
(7 Pairs of Data X 6
Raters)
FIRST REVISION
42 Consistency
Ratings (7 Pairs of
Data X 6 Raters)
Final REVISION
42 Consistency
Ratings (7 Pairs of
Data X 6 Raters)
Statistical Comparisons of Consistency Ratings at BASELINE vs. FIRST REVISION,
BASELINE vs. SECOND REVISION, and FIRST REVISION vs. SECOND REVISION
Figure 1 Schematic of methods to assess extent of data extraction consistency
Hernandez et al.: Criteria and Instructions for Consistent QI ExtractionsJGIM

assessments for the final revision) as displayed in Figure 1.
This non-parametric data on extraction consistency was com-
pared between the groups (baseline vs. first revision, baseline
vs. final revision, first revision vs. final revision) using the
related-samples Wilcoxon signed-rank test (SPSS). We pro-
vide mean and median extraction consistency values for base-
line, first revision, and final revision and a P value of < 0.05
considered statistically significant. The final instruction set for
the criteria are included in the Appendix.
Assessing the Relative Value of the Criteria
Two quality improvement clinicians from Hartford Healthcare
and the University of Connecticut Health Center were first
asked to rate how difficult it is to find and use quality im-
provement studies i n heal th system quality impro vement
endeavors. They were then asked how valuable it would be
if AHRQ found and collated the background information
about the interventions and the health systems from published
quality improvement studies. For both questions, people were
asked to use a 0 to 10 rating scale where 0 denotes no difficulty
or no value and 10 denotes great difficulty or great value,
respectively. While this is a subjective, and therefore imper-
fect, rating system, it does provide some insight into some-
thing that is very difficult to quantify otherwise. Our quality
improvement clinicians (identified in the Acknowledgments
section) have years of experience in quality improvement and
come from both a small rural academic medical center and a
large, urban and suburban, community health system to max-
imize applicability.
RESULTS
Impact of Iterative Instruction Revision on
Consistency
For our primary aim, Table 2 delineates the degree to which
raters found similarities in data extracted from studies for each
of the randomly selected criteria between our two independent
extractors. At baseline, we found very little extraction similar-
ity for the criteria in the source studies by our two independent
systematic reviewers. After we refined the instructions about
how to extract data for the criteria the first time, we dramati-
cally improved the consistency of extraction scores between
the two reviewers by 5.2-fold (P < 0.001). The second and
final revision of the instructions again increased the rating of
similarity between the two reviewers versus baseline by 5.8-
fold ( P < 0.001) but only nominally different from that
achieved after the first revision (P = 0.14) (Table 2). The mean
and median agreement scores of 6.81 and 7 represent a mod-
erate level of agreement with the final set of instructions for
extracting data for these criteria.
For example, at baseline for the criterion #7 “Duration of
Intervention,” reviewer A extracted “Duration of intervention:
two face-to-face group meetings lasting ~2.5 h each. Length of
time of intervention: 2 years.” and reviewer B extracted “2-3
weeks, 4 months.” After the first revision, both reviewers
extracted “Duration of intervention: Three 1-day learning
sessions. Length of intervention: 12 months” and “Duration
of the intervention: 3 1-day sessions; length: 12 months,”
respectively. Finally, after the final revision, both reviewers
extracted: “Duration of intervention: First component (Five 3-
h sessions over 5 months, plus 2 additional 3-h sessions at end
of 1st year); second component (3-h in first year); third com-
ponent (no time specified for monthly visits up to 2y fup).
Total for first and second component was 24 h; unknown for
third component. Length of intervention: 2 years.” and “First
and fourth component: 3 hours x 7=21 hours; second compo-
nent: 3 hours; third component: duration of intervention not
specified. Length of intervention: 2 years.” respectively.
Assessing the Value of the Criteria
The two health system representatives rated how difficult it is
to find and use quality improvement studies in health system
quality improvement endeavors in general. On a scale of 0 to
10, the first key informant rated it a 6 (moderately difficult)
while the second reviewer rated it a 4 (moderately difficult),
justified by the paucity of published quality improvement
literature that they can locate and the time involved in evalu-
ating them. They were then asked how valuable it would be if
AHRQ found and collated the demographic information about
the health systems and the interventions used in published
quality improvement studies. On a scale or 0 to 10, the first
key informant rated it a 9 (highly valuable) and the second
rated it a 6 (moderately valuable).
DISCUSSION
This is the first study, which we are aware of, that explicitly
looked at the consistency of data extraction from quality
improvement studies. Our systematic reviewers are trained
members of EPCs with ample experience but in the absence
of detailed instructions, the consistency of data extraction from
quality improvement studies in our study was very poor.
Fortunately, we found that heterogeneous data extraction is
surmountable with explicit instructions developed in an itera-
tive fashion. We went through two refinements of the data
extraction instructions for each criterion and were able to
improve the consistency of extraction from baseline to the
Table 2 Data Extraction Consistency Ratings
Mean (standard
deviation)
Median (25th–75th
percentile)
Baseline ratings 1.17 (1.85) 0 (0–3)
First revision
ratings
6.07 (2.76)* 6 (3–9)
Final revision
ratings
6.81 (1.94)*
,
7(6–8)
*P < 0.0001 vs. baseline, P = 0.14 vs. first revision. Scores could range
from 0 to 10 where 0 = no consistency of extraction between systematic
reviewers and 10 = perfect consistency
Hernandez et al.: Criteria and Instructions for Consistent QI Extractions JGIM

final revision from 1.17 to 6.81 out of 10. Since the second
refinement only increased the consistency of extraction slight-
ly over the first, further refinements are unlikely to provide
appreciable enhancements. We believe that the uniqueness of
quality improvement studies requires this standardized ap-
proach to data extraction versus more traditional observational
studies and randomized trials. Since we only looked at one
disease state and a relatively few number of studies, there are
limitations to our approach and perhaps some issues of appli-
cability as well. As such, further research looking at other
disease states would be beneficial.
Our criteria overlaps with that of the Quality Improvement -
Minimum Quality Criteria Set (QI-MQCS), a pared down
version of the criteria created by the Standards for Quality
Improvement Reporting Excellence (SQUIRE) group.
10, 14
The QI-MQCS tool, developed with input from nine expert
panelists, selected 14 criteria that the panelists gave a mean
rating of 2.0 or greater in terms of importance (scale from 1 to
3 where 3 denoted it should be included, 2 denoted it may be
included, and 1 denoted it should not be included) and two
additional criteria not vetted through the expert panel. Of the
16 QI-MQCS criteria, our criterion set includes 12 of them
(organizational motivation, organizational readiness, interven-
tion, intervention rationale, organizational characteristics, im-
plementation, timing, adherence/fidelity, penetration or reach,
sustainability, comparator, and data source). Our criteria did
not include the study design, health outcomes, ability for the
intervention to be replicated, or inclusion of study limitations
criteria. However, our quality improvement applicability table
would accompany the standard information presented in EPC
evidence reviews where the study design, health outcomes,
and qualitative or quantitative synthesis of the results appear.
In total, our criterion encompasses and expands on their crite-
ria. This is not surprising since we both relied on the SQUIRE
2.0 criteria while we also used other sources to identify criteria
that we felt were valuable.
10–13
Testing the criterion instructions using quality improvement
instructions in other diseases is a valuable next step as is more
fully vetting the 33 criteria for their usefulness and
completeness.
CONCLUSIONS
Our study suggests that learning health systems need
support in identifying quality improvement studies and
the key features of the interventions and the institutions
that carried them out. In the absence of explicit and
detailed instructions, there is very high heterogeneity in
data extraction among independent reviewers that
improves considerably with t he refinement of the criteria
using an explicit process. Now that consistency of ex-
traction has been enhanced for each of our candidate
criteria, a future study should determine the relative
value of each criterion to learning health systems.
Acknowledgments:
We acknowledge Christina M. Polomoff, Pharm.D., BCACP, BCGP,
from Integrated Care Partners, Hartford Healthcare’sphysician-led
clinically integrated network, and Kevin Chamberlin, Pharm.D.,
FASCP, from UConn Health’s John Dempsey Hospital for their
contributions as health system key informants.
Role of the Funder: A representative from AHRQ served as a Con-
tracting Officer’s Technical Representative and provide technical assis-
tance during the conduct of the full evidence report and provided com-
ments on draft versions of the full evidence report. AHRQ did not
directly participate in the literature search, determination of study
eligibility criteria, data analysis or interpretation, or preparation, re-
view, or approval of the manuscript for publication.
Corresponding Author: C. Michael White, Pharm. D., FCP, FCCP;
University of Connecticut School of Pharmacy, Storrs, CT, USA
(e-mail: Charles.white@uconn.edu).
Funding Information This project was funded under Contract No.
HHSA290-2015-00012I Task Order I from the Agency for Healthcare
Research and Quality (AHRQ), U.S. Department of Health and Human
Services (HHS).
Compliance with Ethical Standards:
Conflict of Interest: The authors declare that they do not have a
conflict of interest.
Disclaimer: The authors of this manuscript are responsible for its
content. Statements in the manuscript do not necessarily represent
the official views of or imply endorsement by AHRQ or HHS.
REFERENCES
1. The Joint Commission. Quality Accreditation Standards Information.
Available at: https://www.jointcommission.org/standards_information/
jcfaq.aspx. Accessed, 8/6/18.
2. Sullivan T. According to CMS 1,500 hospitals will receive bonus pay-
ments in 2020. December 4, 2019. Available at: https://www.policymed.
com/2019/12/according-to-cms-1500-hospitals-will-receive-bonus-pay-
ments-in-2020.html Accessed 2/21/20.
3. Health Services Advisory Group. National Impact Assessment of the
Centers for Medicare & Medicaid Services (CMS) Quality Measures
Report. Feb 28, 2018. Available at: https://www.cms.gov/Medicare/
Quality-Initiatives-Patient-Assessment-Instruments/QualityMeasures/
Downloads/2018-Impact-Assessment-Report.pdf. Accessed 8/6/18.
4. Institute of Medicine. 2013. Best Care at Lower Cost: The Path to
Continuously Learning Health Care in America. Washington, DC: The
National Academies Press. Available at: https://doi.org/10.17226/
13444. Accessed. 8/6/18
5. The Learning Healthcare Project. Available at: http://www.learning-
healthcareproject.org/section/background/learning-healthcare-system.
Accessed 8/6/18.
6. McGinnis JM Evidence-based medicine - engineering the Learning
Healthcare System. Stud Health Technol Inform. 2010;153:145-57.
7. White CM, Sanders-Schmidler GD, Butler M, et al. Understanding
health systems’ use of and need for evidence to inform decisionmaking.
Agency for Healthcare Research and Quality (US); 2017. Report 17(18)-
EHC035-EF. Rockville, MD
8. AHRQ Quality Improvement Working Group. Synthesizing Evidence for
Quality Improvement. Agency for Healthcare Research and Quality.
August 8, 2018. Available at: https://effectivehealthcare.ahrq.gov/
topics/health-systems/quality-improvement. Accessed 6/10/2019.
9. Bravata DM, Sundaram V, Lewis R, Gienger A, Gould MK, McDonald
KM, Wise PH, Holty J-EC, Hertz K, Paguntalan H, Sharp C, Kim J,
Wang E, Chamberlain L, Shieh L, Owens DK. Asthma Care. Vol 5 of:
Shojania KG, McDonald KM, Wachter RM, Owens DK, editors. Closing
the Quality Gap: A Critical Analysis of Quality Improvement Strategies.
Hernandez et al.: Criteria and Instructions for Consistent QI ExtractionsJGIM

Technical Review 9 (Prepared by the Stanford University-UCSF Evidence-
based Practice Center under Contract No. 290-02-0017). AHRQ Publica-
tion No. 04(07)-0051-5. Rockville, MD: Agency for Healthcare Research
and Quality. January 2007.
10. Ogrinc G, Davies L, Goodman D, Batalden P, Davidoff F, Stevens D.
Standards for Quality Improvement Reporting Excellence 2.0: revised
publication guidelines from a detailed consensus process. J Surg Res
2016;200(2):676–682.
11. Pinnock H, Barwick M, Carpenter CR for the StaRI Group, et al.
Standards for Reporting Implementation Studies (StaRI): explanation and
elaboration document. BMJ Open. 2017;7:e013318. https://doi.org/10.
1136/bmjopen-2016-013318.
12. Hoffmann T, Glasziou P, Boutron I, et al. Better reporting of inter-
ventions: template for intervention description and replication (TIDieR)
checklist and guide. BMJ 348: g1687. 2014.
13. Guise J-M, Butler ME, Chang C, Viswanathan M, Pigott T, Tugwell P.
AHRQ series on complex intervention systematic reviews—paper 6:
PRISMA-CI extensio n statement an d checklist. J Clin Epidemiol.
2017;90:43–50.
14. Hempel S, Shekelle PG, Liu JL, et al. Development of the Quality
Improvement Minimum Quality Criteria Set (QI-MQCS): a tool for critical
appraisal of quality improvement intervention publications. BMJ Qual
Saf 2015;24:796-804.
Publisher’sNote:Springer Nature remains neutral with regard to
jurisdictional claims in published maps and institutional affiliations.
Hernandez et al.: Criteria and Instructions for Consistent QI Extractions JGIM
